
Your graft cost $847. Medicare paid nothing. The denial reason? “Procedure code inconsistent with product code.” One digit difference between a CPT and a C-code pathway cost you the entire product AND the application fee in a single encounter. That is not a paperwork problem. That is a revenue problem — and it is happening in wound care practices across the country every billing cycle.
Skin substitute billing codes are unforgiving. You are not filling out a simple superbill. Every claim chains together a wound diagnosis, a CPT application code, a product-specific HCPCS code, a cost-group classification, LCD medical necessity requirements, and setting-specific Medicare payment logic. Each link is a potential break point. Miss one and your clean claim becomes an error claim — or worse, an audit target.
2026 raised the stakes further. CMS restructured how skin substitute products are paid in the non-facility setting under the CY 2026 Medicare Physician Fee Schedule Final Rule. The WISeR prior authorization pilot went live January 1, 2026 in six states. New Q-codes entered the system, cost-group assignments shifted, and Medicare Advantage plans tightened their documentation review timelines. Practices that updated their workflows are collecting. Practices that did not are watching denial rates climb.
The good news: every one of those rules is knowable. This guide gives you the complete 2026 framework — CPT codes 15271–15278, A-codes, Q-codes, HCPCS updates through January 2026, high-cost vs low-cost group logic, Medicare setting differences, ICD-10 linkage, documentation requirements, modifier rules, and prior authorization strategy — all in one place. By the time you finish reading, your team will code faster, document cleaner, and sleep better on audit night.
Direct Answer — What Are Skin Substitute Billing Codes? Skin substitute billing codes consist of two code types filed on every claim: a CPT procedure code (15271–15278 for high-cost products or C5271–C5278 for low-cost products in facility settings) that reports the application procedure, and an HCPCS product code (an A-code for synthetic/device-cleared products or a Q-code for biologic/tissue-based products) that reports the specific graft used. Both codes must appear on the same claim, on the same date of service, matched to the correct wound site, wound size, and CMS cost-group classification. Filing either code alone will result in denial.
→ Talk to Our Wound Care Billing Specialists — Get a Free Billing Audit
How Much Revenue Is
Your Practice Leaving Behind?
Enter your monthly graft volume, denial rate, average product cost, and payer mix. See your current revenue leakage, 2026 WISeR PA overhead impact, recovery opportunity, and annual gap — in real time.
What Are Skin Substitute Billing Codes?
Every denied graft claim has a root. Most of the time, that root is a misunderstanding of what a skin substitute claim actually looks like at the code level.
The Centers for Medicare & Medicaid Services classifies skin substitutes as cellular and/or tissue-based products (CTPs) — not as wound dressings, not as supplies, and not as simple biologics that get tossed into a miscellaneous supply bucket. CMS and Local Coverage Determinations like LCD L36690 distinguish CTPs from non-graft wound dressings specifically because the billing logic is completely different. Regular moisture-retentive dressings, foam dressings, and hydrocolloids are included in standard wound care management. Skin substitute graft procedures are separately reported using anatomic site and surface area — a fundamentally different mechanism.
Three distinct code families drive every skin substitute claim:
| Code Type | Purpose | Common Example |
|---|---|---|
| CPT Application Code | Reports the procedure of applying the graft — site, wound size, fixation | 15271 (trunk/arms/legs, first 25 sq cm) |
| HCPCS A-Code | Reports a synthetic or 510(k)-cleared product | A2002 Mirragen Advanced Wound Matrix |
| HCPCS Q-Code | Reports a biologic or tissue-based product | Q4101 Apligraf |
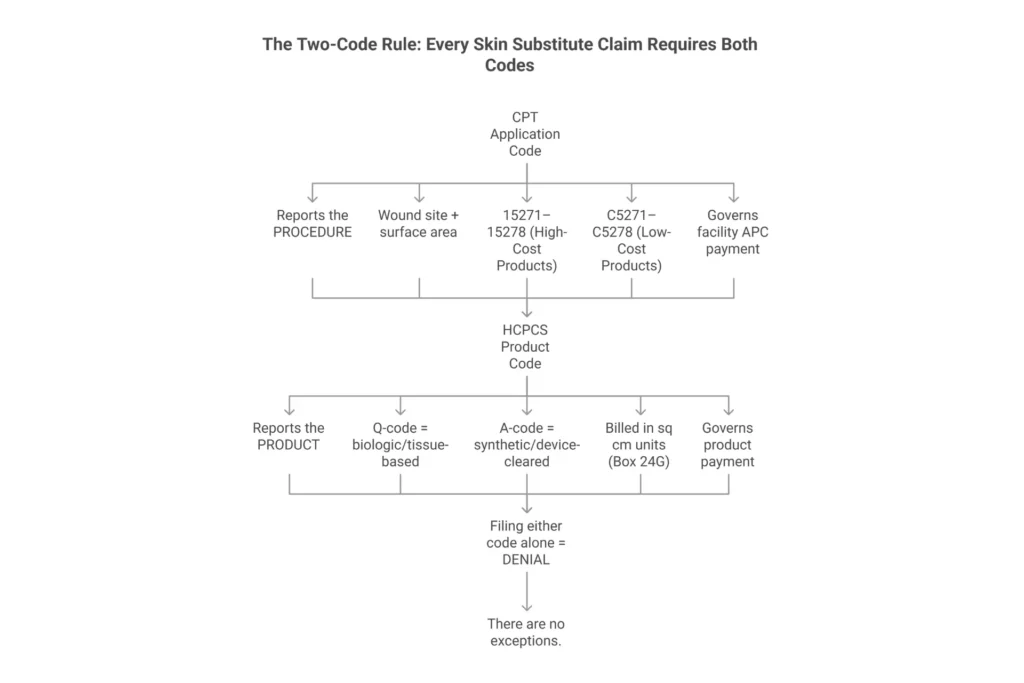
Here’s what most practices miss: neither code stands alone. The two-code rule is not a billing preference — it is a structural requirement. Noridian’s wound care guidance explicitly states that skin substitute product codes must be filed with a same-claim application procedure code that matches the product’s cost-group classification. File the application code without the product code and Medicare has no way to price the product. File the product code without the application code and the claim has no procedural anchor and will deny.
The split also determines how Medicare pays. The application code (the CPT or C-code) governs the facility payment and APC assignment. The HCPCS product code governs how the product itself is priced. In the hospital outpatient setting, high-cost products get additional separate payment consideration on top of the procedure APC. Low-cost products are packaged into the procedure payment. Get the pairing wrong — for example, using a low-cost C-code with a high-cost product — and you can lose hundreds of dollars per claim.
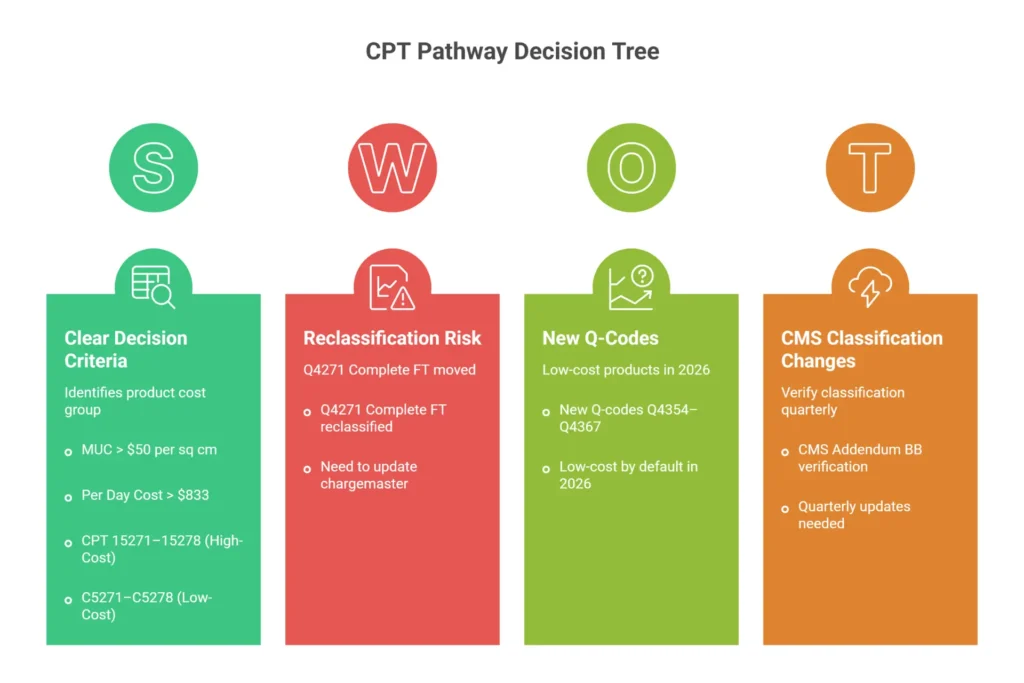
One critical 2026 change: Under the CY 2026 MPFS Final Rule, CMS restructured the non-facility payment model for skin substitute products applied in the physician office setting. Practices that relied on the previous separate product reimbursement structure for office-based graft applications must verify their current MPFS payment methodology and update billing workflows accordingly before submitting any claims for dates of service on or after January 1, 2026.
Want help setting up your claim workflow correctly for 2026? Our wound care billing specialists configure your billing for every code pairing, cost group, and setting under the 2026 rules. Learn about our wound care RCM services →
Skin Substitute CPT Codes 15271–15278: The Complete 2026 Breakdown
Here is the first place revenue gets lost: picking the wrong code family for the anatomical site, then compounding it by billing based on product sheet size instead of measured wound area.
CPT codes 15271–15278 report the application of skin substitute grafts using two variables: anatomical site and wound surface area. The AMA groups these codes into two families based on site complexity, then subdivides them by whether the total wound surface area is under 100 square centimeters or at least 100 square centimeters. These CPT code descriptors themselves did not change in 2026, but the payment rates, OPPS APC assignments, and non-facility reimbursement pathways are updated under the CY 2026 Final Rules.
| CPT Code | Description | Anatomic Group | Wound Size Logic | 2026 OPPS APC | 2026 ASC Rate |
|---|---|---|---|---|---|
| 15271 | Application, first 25 sq cm or less | Group 1: Trunk, arms, legs | Total area < 100 sq cm | APC 5054 | G2 rate — verify Addendum B |
| +15272 | Each additional 25 sq cm | Group 1 | Add-on to 15271 | Packaged | Packaged |
| 15273 | Application, first 100 sq cm | Group 1: Trunk, arms, legs | Total area ≥ 100 sq cm | APC 5055 | G2 rate — verify Addendum B |
| +15274 | Each additional 100 sq cm | Group 1 | Add-on to 15273 | Packaged | Packaged |
| 15275 | Application, first 25 sq cm or less | Group 2: Face, scalp, neck, hands, feet, genitalia, multiple digits | Total area < 100 sq cm | APC 5054 | G2 rate — verify Addendum B |
| +15276 | Each additional 25 sq cm | Group 2 | Add-on to 15275 | Packaged | Packaged |
| 15277 | Application, first 100 sq cm | Group 2: Same special sites | Total area ≥ 100 sq cm | APC 5055 | G2 rate — verify Addendum B |
| +15278 | Each additional 100 sq cm | Group 2 | Add-on to 15277 | Packaged | Packaged |
Always verify current rates against the CY 2026 OPPS Addendum B and Addendum D1 before billing. OPPS rates are subject to geographic CBSA wage-index adjustment and the 2% Medicare sequestration reduction. CMS updates quarterly addenda in January, April, July, and October — lock your chargemaster rates to the applicable quarter, not just the annual final rule.
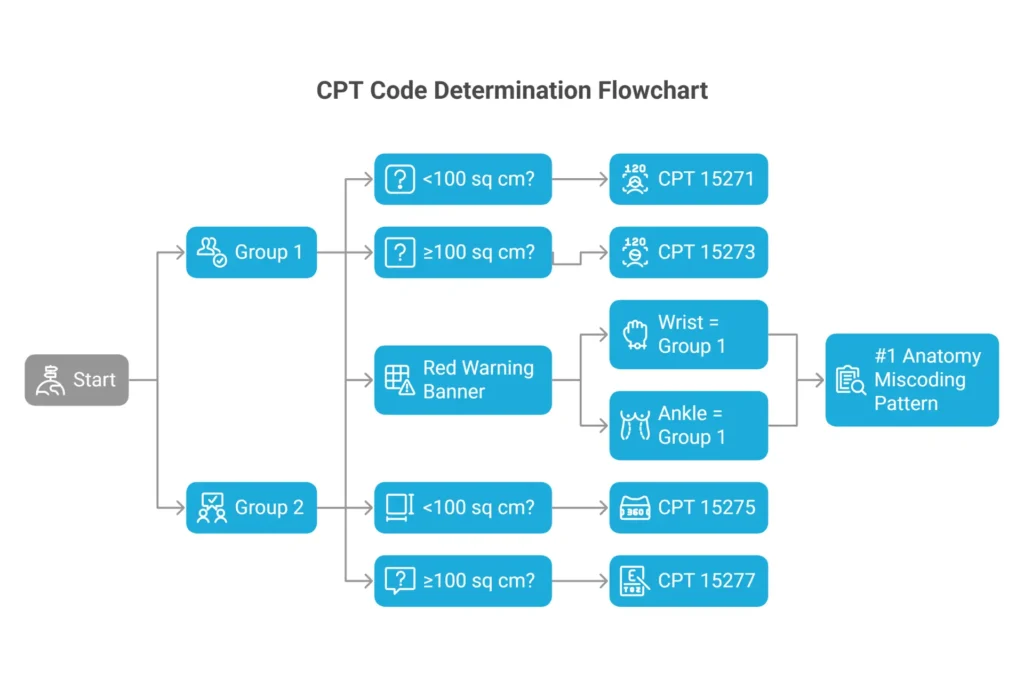
The Anatomy Trap That Triggers Audits
Group 1 includes the trunk, arms, and legs. Group 2 includes the face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, and multiple digits. Here is the trap that trips practices constantly: the wrist codes as an arm (Group 1), not a hand (Group 2). Similarly, the ankle codes as a leg (Group 1), not a foot (Group 2). CPT coding logic follows anatomical regions, not surgical specialty convention. A wound on the dorsal wrist is 15271, not 15275. A wound on the lateral malleolus is 15271, not 15275. MAC auditors specifically target the wrist/hand and ankle/foot miscoding pattern in skin substitute reviews — this pattern appeared in multiple MAC Comparative Billing Reports through 2025 and remains an active 2026 audit focus.
How to Calculate Which Code to Use
- Identify the anatomical group. Is the wound on Group 1 or Group 2 sites?
- Measure the wound recipient bed. Use length × width in centimeters. This is the wound — not the product sheet you opened.
- Add contiguous or adjacent wound areas within the same group. Multiple small wounds on the same leg aggregate into one total for code selection.
- Determine the threshold. Total area under 100 sq cm → start with 15271 or 15275. Total area 100 sq cm or greater → start with 15273 or 15277.
- Calculate add-on units. Divide remaining area by 25 (for 15272/15276) or 100 (for 15274/15278). Round any partial unit up — the “or part thereof” rule means even 1 extra square centimeter gets a full add-on unit.
Worked Example: 65 sq cm Diabetic Foot Ulcer, Right Foot
- Site: Right foot. That is Group 2.
- Total wound area: 65 sq cm. Under 100 sq cm, so start with 15275.
- First 25 sq cm = 1 unit of 15275.
- Remaining 40 sq cm ÷ 25 = 1.6, rounds up to 2 units of +15276.
- Correct claim: 15275 × 1 unit, +15276 × 2 units.
- Product code (e.g., Q4186 EpiFix): 65 units in Box 24G.
Do not let the team bill 3 units of +15276 because the product sheet was 4×4 cm (16 sq cm) applied twice. You bill the wound, not the packaging. Billing product size instead of wound size is one of the most common audit triggers in skin substitute claims — and one of the easiest for MAC post-payment reviewers to spot by comparing procedure note measurements against billed HCPCS units.
What Is Bundled into 15271–15278?
Simple wound bed preparation and routine cleaning are included in these codes. You cannot separately bill CPT 11042–11047 (active wound debridement) for the same wound on the same application day — NCCI edits treat that combination as mutually inclusive for the same wound. You also cannot separately bill CPT 15002 or 15004 (surgical preparation codes) on the same wound and same date.
What about same-day E/M? Modifier -25 on the evaluation and management code is the only path to separately billing an E/M if the visit involved significant separately identifiable work beyond the graft procedure itself. Routine graft monitoring is included. A genuinely separate clinical assessment — like managing uncontrolled diabetes or evaluating a different wound — can support a -25 modifier with proper documentation.
For a complete overview of all wound care billing codes — including debridement, hyperbaric oxygen, and E/M pairing rules — visit our wound care billing codes guide.
HCPCS Product Codes for Skin Substitutes: A-Codes, Q-Codes & 2026 Updates
The product code is where claims hit a wall most often at the chargemaster level. Practices pull a stale code list from last year, use a deleted code, or use a code that moved from one cost group to another — and suddenly every claim for that product is denying.
A-codes primarily cover synthetic, polymer-based, or 510(k)-cleared products. Q-codes primarily cover biologic, human-derived, animal-derived, or tissue-based products regulated under different FDA pathways. Both report product identity to CMS and must appear on the same claim as the application procedure code. The unit for both is typically per square centimeter of wound area treated.
Top Commonly Billed Skin Substitute Product Codes (2026)
| Product | HCPCS Code | 2026 Cost Group | Notes |
|---|---|---|---|
| Apligraf | Q4101 | High-cost | Organogenesis; bilayered living skin construct |
| OASIS Wound Matrix | Q4102 | Low-cost | Smith+Nephew; porcine small intestine submucosa |
| Integra Bilayer Wound Matrix | Q4104 | High-cost | Integra LifeSciences |
| Dermagraft | Q4106 | High-cost | Organogenesis; cryopreserved human fibroblast-derived dermis |
| AmnioFix | Q4115 | High-cost | MiMedx; amniotic membrane allograft |
| EpiFix (sheet form) | Q4186 | High-cost | MiMedx; dehydrated human amnion/chorion membrane |
| Kerecis Omega3 Wound | Q4158 | High-cost | Kerecis; fish-skin graft |
| AmnioCore | Q4264 | High-cost | MiMedx |
| Complete FT | Q4271 | High-cost | Reclassified high-cost April 2025; verify 2026 Addendum BB |
| Mirragen Advanced Wound Matrix | A2002 | Verify current Addendum BB | ETS Wound Care; synthetic bioabsorbable matrix |
Always verify current cost-group status against the CY 2026 CMS quarterly Addendum BB. Cost-group assignments change quarterly. See: CMS Quarterly Addenda Updates
The 2026 Q-Code Landscape — What Carried Over and What Changed
CMS Change Request 14017 (effective April 1, 2025) established 14 new Q-codes (Q4354–Q4367) as low-cost defaults. All 14 of those codes carry into 2026 still classified as low-cost unless CMS has accumulated pricing data to support reclassification. Before billing any of them, confirm current cost-group status in the 2026 Addendum BB — default low-cost classification is not permanent.
Q4231 (Corplex P) remains deleted as of March 31, 2025. Any claim submitted with Q4231 for a date of service from April 2025 onward will hard-stop reject. If your chargemaster was not updated in 2025, correct it immediately.
Q4271 (Complete FT) remains high-cost as of January 1, 2026. The reclassification that took effect April 1, 2025 is still in force. This product pairs with CPT 15271–15278, not C5271–C5278.
January 2026 HCPCS update cycle: CMS releases the annual HCPCS Level II code set effective January 1 each year. New skin substitute products approved through the HCPCS application process in 2025 will receive new Q-codes effective January 1, 2026. Verify the complete January 2026 HCPCS release at cms.gov/Medicare/Coding/HCPCSReleaseCodeSets against your full product formulary before billing in any quarter of 2026.
Here’s what most practices miss: the quarterly update cycle is the enemy of stale code lists. CMS updates the HCPCS code set in January, April, July, and October. A chargemaster audit every quarter is not optional if you are billing skin substitutes at volume. Bookmark the CMS Quarterly Addenda Updates page and assign a specific staff member accountability for this review each quarter.
For amniotic membrane products specifically — including EpiFix, AmnioFix, and AmnioCore — we have a complete breakdown in our amniotic membrane graft billing guide.
High-Cost vs Low-Cost Skin Substitute Groups: How CMS Determines 2026 Payment
This is the most financially consequential section in the entire article. Read it carefully. One wrong classification can generate hundreds of dollars in under-payment per claim or put your practice in an overpayment recoupment situation.
CMS splits skin substitute products into two groups for OPPS and ASC payment purposes:
- High-cost group: Mean Unit Cost (MUC) above $50 per square centimeter OR Per Day Cost (PDC) above $833. These products pair with CPT 15271–15278 in facility billing.
- Low-cost group: Products that do not meet either threshold, and all new products by default until pricing data supports reclassification. These products pair with HCPCS C5271–C5278 in facility billing.
That pairing distinction is not a formatting preference. It is the structural mechanism CMS uses to determine how the application procedure is priced. High-cost products trigger a different reimbursement pathway. Low-cost products are packaged differently. Put the wrong application code family with the wrong product and the claim either underpays or triggers audit flags for overbilling.
C5271–C5278: The Low-Cost Application Code Family
These codes mirror the CPT 15271–15278 family in structure — same anatomical groups, same wound-size increments. The only difference is they are specifically designated for low-cost skin substitute products in facility settings:
| Low-Cost Code | Mirrors | When to Use |
|---|---|---|
| C5271 | 15271 | Low-cost product, Group 1, first 25 sq cm (total area <100 sq cm) |
| +C5272 | +15272 | Add-on, Group 1, each additional 25 sq cm |
| C5273 | 15273 | Low-cost product, Group 1, first 100 sq cm (total area ≥100 sq cm) |
| +C5274 | +15274 | Add-on, Group 1, each additional 100 sq cm |
| C5275 | 15275 | Low-cost product, Group 2, first 25 sq cm (total area <100 sq cm) |
| +C5276 | +15276 | Add-on, Group 2, each additional 25 sq cm |
| C5277 | 15277 | Low-cost product, Group 2, first 100 sq cm (total area ≥100 sq cm) |
| +C5278 | +15278 | Add-on, Group 2, each additional 100 sq cm |
What the 2026 Non-Facility Payment Change Means for Your Practice
The CY 2026 MPFS Final Rule restructured how skin substitute products are separately reimbursed in the physician office (non-facility) setting. Under the prior model, many high-cost products received separate payment based on the product code alongside the application CPT code on the same MPFS claim. The 2026 changes altered the packaging and separate-payment logic for office-based skin substitute claims.
Here’s what most practices miss: this change affects the office setting specifically — not HOPD OPPS billing. If your practice applies skin substitutes in a hospital outpatient wound clinic, your OPPS payment pathway remains governed by the APC system and the high-cost/low-cost cost group structure described throughout this article. If you apply skin substitutes in an independent physician office, verify your current MPFS payment structure for every product in your formulary before submitting 2026 dates of service. Contact your MAC directly or your billing service for setting-specific guidance.
The Q4271 Reclassification — Still in Force for 2026
Before April 1, 2025, Complete FT (Q4271) was low-cost. Practices using Q4271 were billing C5271 or C5275 as the application code — completely correct for that period.
Effective April 1, 2025, Q4271 moved to the high-cost group. That reclassification carries into 2026. Starting that day in 2025 and continuing through all 2026 dates of service, the correct application code is CPT 15271 or 15275 — not C5271 or C5275. Any facility that still has Q4271 mapped to a C-code in its chargemaster is filing procedure-device mismatches on every claim.
Revenue impact of the wrong pairing: a $600 high-cost graft applied to a 25 sq cm wound billed through the low-cost C-code pathway can result in the practice absorbing the product cost entirely. That gap can exceed $500 per encounter on some high-cost grafts.
Here’s what most practices miss: pass-through status is not the same as high-cost status. A product with temporary CMS pass-through classification (Status Indicator K) can receive separate payment that bypasses normal packaging logic during that limited window. Once pass-through ends, the product defaults back to its cost-group assignment. Never assume a new premium product entering the market is automatically high-cost.
For questions about how your specific product list maps to the current 2026 cost groups, our medical coding specialists can audit your chargemaster against the current CMS Addendum BB in one business day.
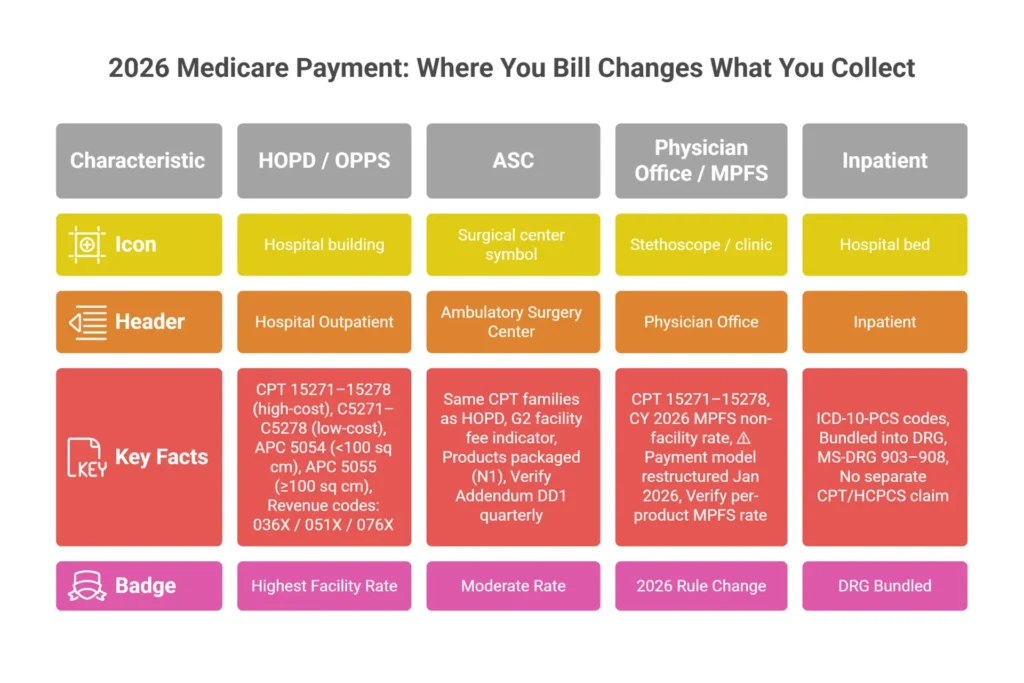
Medicare Billing for Skin Substitutes by Clinical Setting — 2026 Update
Where you apply the graft changes how Medicare pays for it. This is not a subtle difference. The same CPT code can generate dramatically different revenue depending on the clinical setting — and that gap widened under the CY 2026 payment rules.
Setting Comparison at a Glance
| Setting | Application Codes Used | 2026 Payment Reference | Key Rules |
|---|---|---|---|
| HOPD / OPPS | CPT 15271–15278 (high-cost) or C5271–C5278 (low-cost) | APC 5054, APC 5055 — verify CY 2026 Addendum B | Revenue codes 036X, 051X, 076X; PO/PN modifiers for off-campus |
| ASC | Same CPT families | G2 facility fee — verify CY 2026 Addendum DD1 | Products packaged (N1) unless pass-through; wage-index adjusted |
| Physician Office / MPFS | CPT 15271–15278 | CY 2026 MPFS non-facility rate — restructured Jan 1, 2026 | Significant payment model change effective 2026; verify before billing |
| Inpatient | ICD-10-PCS codes / DRG bundling | Bundled into DRG (MS-DRG 903–908) | No outpatient CPT/HCPCS product claims |
Hospital Outpatient (HOPD) Under CY 2026 OPPS
HOPD remains the most predictable outpatient setting for skin substitute procedure payment. CMS assigns the core application procedures to APC 5054 for the under-100 sq cm family and APC 5055 for the 100+ sq cm family. These APCs carry into 2026 under the CY 2026 OPPS Final Rule — verify the national unadjusted rates in the published Addendum B, then apply your CBSA wage index and the 2% sequestration reduction for actual expected payment.
Revenue code selection matters. Wound care treatment rooms typically bill 076X, hospital-based wound clinics use 051X, and surgery suites use 036X. Using the wrong revenue code does not always deny outright, but it can trigger edit failures and payment delays. Confirm your charge description master maps each wound care location to the correct revenue code family.
Off-Campus Provider-Based Departments: The Modifier You Cannot Ignore
If your wound care clinic opened or started billing as a hospital outpatient department after November 2, 2015, it is a non-excepted off-campus provider-based department. Use modifier -PN on every applicable claim. Payment is calculated at the MPFS office-equivalent rate — significantly lower than full OPPS APC rates.
If the clinic was already billing as a hospital outpatient department on or before November 2, 2015, it is excepted. Use modifier -PO. Full OPPS rates apply.
Here’s what most practices miss: billing -PO when you should be billing -PN is an overpayment. MAC post-payment reviews specifically target PBD modifier patterns. Getting this right is not optional.
ASC Billing in 2026
ASC facility fees come through the G2 payment indicator. Product costs are typically packaged as N1 (no separate payment) for products without current pass-through status in the ASC setting. Check the current CY 2026 ASC Addendum DD1 for applicable G2 and P3 indicators by product code. New products that received CMS pass-through status in 2025 may carry that status into part of 2026 before transitioning back to packaging.
The CY 2026 MPFS Non-Facility Setting Change
This is the most operationally disruptive 2026 payment change for wound care practices that apply skin substitutes in the independent office setting. CMS restructured how skin substitute products are separately paid under the MPFS for non-facility dates of service on or after January 1, 2026. If your practice relies on office-based graft application as its primary care setting, you need to verify the exact payment mechanism for each product in your formulary under the 2026 MPFS rules before submitting any claims. Contact your MAC for setting-specific written guidance and update your practice management system to reflect the correct 2026 fee schedule structure.
Medicare Advantage — The Front-End Friction Layer
Traditional Medicare Part B now has an active prior authorization requirement in WISeR pilot states (see H2 #9). Medicare Advantage plans have always required PA or utilization review, and that requirement is tighter in 2026. UHC Medicare Advantage Policy PCA-1-24-02596 uses a post-service, pre-payment review model. Non-porcine products trigger an enhanced clinical review process. The documentation package must reflect 4+ weeks of conservative care, wound measurements, and less than 50% closure before the graft.
For a complete breakdown of Medicare coverage rules for wound grafts, see our Medicare wound care billing guide and our wound graft Medicare coverage overview.
ICD-10 Codes for Skin Substitute Claims: Medical Necessity Starts Here
A perfectly coded claim with the right CPT, the right HCPCS, and the right cost-group pathway still fails if the diagnosis does not support medical necessity. MAC auditors reviewing skin substitute claims look at diagnosis codes to verify that the wound type, etiology, and severity actually qualify for graft coverage under the applicable LCD.
ICD-10 Code Reference by Wound Type
| Wound Type | Primary ICD-10 Code(s) | Clinical Notes |
|---|---|---|
| Diabetic foot ulcer (Type 2) | E11.621 (foot); E11.622 (other skin ulcer) | Requires laterality; pair with Z79.4 for insulin use |
| Diabetic foot ulcer (Type 1) | E10.621 (foot); E10.622 (other) | Same laterality and documentation requirements |
| Venous leg ulcer | I83.001–I83.229 | Right/left/bilateral laterality required; I87.31x for chronic venous hypertension |
| Pressure ulcer (stage 3/4) | L89.xxx (site + stage) | Stage 1 and 2 not covered; unstageable (L89.xx0) covered under most LCDs |
| Chronic non-healing wound | L98.499 | Use only when etiology truly unknown; expect additional scrutiny |
| Surgical wound dehiscence | T81.31XA (superficial); T81.32XA (deep) | XA = initial encounter; update to XD/XS on follow-up |
| Burn wound | T20–T32 series | Full-thickness burns; use correct site, degree, and body-surface percentage codes |
LCD coverage for skin substitute claims is MAC-specific. LCD L35041 (Novitas JL/JM) covers diabetic and pressure ulcers of the lower extremity. LCD L36690 (CGS J15) covers wound application of CTPs broadly. LCD L36377 (First Coast JF/JN) focuses on DFU and VLU of the lower extremities and requires documented adequate arterial flow. All three LCDs share one common requirement: unspecified or non-specific diagnosis codes will result in medical necessity denials.
Laterality is not optional. A venous ulcer coded as I83.009 (unspecified, bilateral) instead of I83.011 (right, lower leg) or I83.012 (left, lower leg) will trigger a denial under any of the major LCDs. The same applies to diabetic ulcers — E11.621 requires you to specify which foot in your documentation, and the code should reflect the actual wound location.
Common ICD-10 Confusion — Quick Answers:
- Z94.89 (other transplanted organ and tissue status) is a secondary code documenting that a patient has a graft in place. It is not a primary diagnosis and does not establish wound medical necessity by itself.
- Z76.9 (encountering health services, unspecified circumstances) is not an acceptable primary diagnosis for skin substitute claims. Using it as the primary code invites an MA130 medical necessity denial.
- There is no ICD-10-CM code that specifically means “skin substitute application.” Your primary diagnosis must be the active wound etiology.
For specific ICD-10 guidance on outpatient wound care encounters, visit our outpatient wound care billing guide.
Documentation Requirements: What Makes a Clean Skin Substitute Claim in 2026
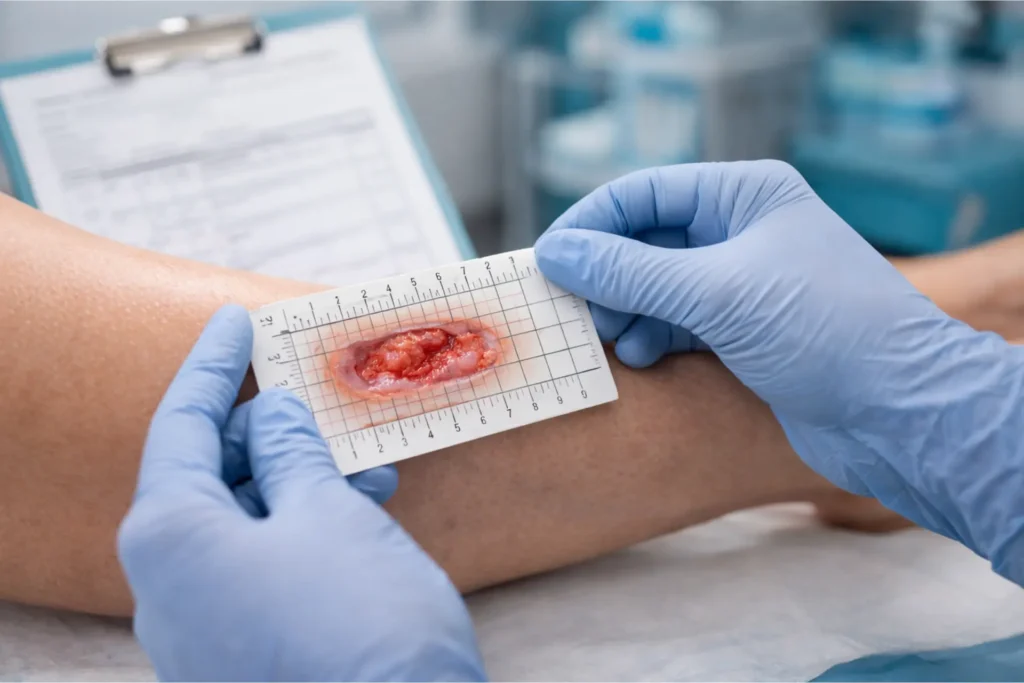
Auditors do not deny claims because providers did bad medicine. They deny claims because the medical record does not prove good medicine was done. Those are different problems, and the second one is entirely preventable.
The 4-Week Conservative Care Requirement
Before applying a skin substitute, most LCDs require documented evidence that standard wound care has failed. For diabetic foot ulcers and venous leg ulcers, that means at minimum 4 consecutive weeks of documented conservative care with weekly clinical visits, each containing:
- Length × Width × Depth measurements in centimeters for every wound
- Wound bed description (granulation percentage, slough, eschar, exudate type and volume)
- Treatment rendered at that visit (debridement, offloading, dressing applied)
- Offloading compliance (DFU) or compression therapy compliance (VLU)
- Wound closure calculation — the wound must show less than 50% reduction in surface area over the 4-week period to qualify for graft use
If a patient misses even one weekly visit during that window, document why. If the wound is healing faster than 50%, a skin substitute is not indicated — and billing one anyway is both a clinical and a compliance problem.
Application Day Documentation
On the day of graft application, your operative note or procedure note must include:
- Pre-application wound measurements (the measurements you bill from)
- Wound bed assessment confirming clean granulating bed, no active infection
- Product name, HCPCS code, lot number, and expiration date
- Product size used and area applied (these can differ; document both)
- Fixation method and materials — sutures, staples, tissue adhesive, or bolster. This is required. LCD L36377 language notes that the graft is anchored using clinician-selected fixation. If your note says the product was “applied to wound” without describing how it was secured, auditors can question whether a true surgical graft was performed or whether a dressing was placed.
Vascular Assessment Documentation
For lower-extremity wounds, document adequate vascular supply. ABI results of 0.7–1.3 indicate adequate perfusion. ABI above 1.3 requires toe pressure testing (toe pressure > 30 mmHg) because non-compressible vessels make ABI unreliable. TcPO2 values above 40 mmHg are also acceptable. These studies should be within 12 months of the graft application date.
Infection Status
Active, untreated infection is a non-coverage condition under most LCDs. Document the absence of clinical infection: no erythema extending more than 2 cm beyond the wound edge, no purulence, patient afebrile, no leukocytosis. If the patient had infection that was treated prior to grafting, document the antibiotic course, duration, and resolution before the application date.
Here’s what most practices miss: EHR copy-paste notes and cloned weekly assessments are audit bait. If your wound clinic has 8 weeks of notes that read identically except for the date, MAC medical reviewers flag that as fabricated or templated documentation. Measurements must change week to week even if small. Clinical assessment must reflect actual observation. Every note should read like the clinician actually looked at this specific wound on this specific date.
ABN Requirements
When a skin substitute service may not be covered by Medicare, issue an Advance Beneficiary Notice (ABN) using Form CMS-R-131 before the service. The ABN protects the provider’s right to bill the patient if Medicare denies. Without a signed ABN, you cannot collect from the patient when Medicare denies for medical necessity. Append modifier -GA to the claim when an ABN is on file.
Modifiers, Bundling Rules & Billing Errors to Avoid
The modifier list for skin substitute billing is not long. The consequences of using the wrong one — or forgetting one — can be significant.
Modifier Quick Reference
| Modifier | Use Case | Clinical Example |
|---|---|---|
| -59 | Distinct procedural service when no X-modifier fits better | Different wound, same body region, insufficient anatomical distinction for -XS |
| -XS | Separate structure — preferred over -59 when sites are anatomically distinct | 15271 on left leg wound + 15275 on left hand wound, same DOS |
| -25 | Significant, separately identifiable E/M on same day as procedure | Managing new systemic issue during graft visit |
| -57 | Decision for major surgery (90-day global period) | Rarely applies to skin substitute — use -25 for minor procedures with 0-day global |
| -51 | Multiple procedures | OPPS handles MPPR automatically; do not append -51 in OPPS contexts |
| -PN | Non-excepted off-campus provider-based department | Hospital-owned wound clinic opened after Nov 2, 2015 |
| -PO | Excepted off-campus provider-based department | Hospital-owned wound clinic already billing before Nov 2, 2015 |
CMS guidance explicitly prefers X{EPSU} modifiers (XS, XE, XP, XU) over modifier -59 when the more specific modifier accurately describes the situation. Modifier -59 is still valid but should be your last choice, not your default.
NCCI Bundling — The Non-Negotiable Edits
The National Correct Coding Initiative (NCCI) Procedure-to-Procedure (PTP) edits treat certain code pairs as inseparable for the same wound on the same date. These are Indicator 0 edits — no modifier overcomes them for the same wound:
- 11042–11047 + 15271–15278 on the same wound, same day → Bundled. Perform and bill your debridement on prior dates during the conservative care phase.
- 15002 + 15271 on the same wound, same day → Bundled. Surgical preparation is included in the application code global.
- 15004 + 15275 on the same wound, same day → Bundled. Same logic for Group 2 sites.
The key word is same wound. Debridement of a completely separate wound at a different anatomical location on the same date may be separately billable using modifier -XS or -59, with documentation clearly distinguishing the two wounds.
MUE Limits — Know the Ceilings Before You Bill
Medically Unlikely Edits (MUEs) set the maximum units CMS considers credible per date of service. Primary codes like 15271 and 15275 have an MUE of 4. Add-on codes like +15272 and +15276 have MUE values of 12. Exceeding these limits without exceptional supporting documentation triggers an automatic hard-stop denial that requires manual appeal.
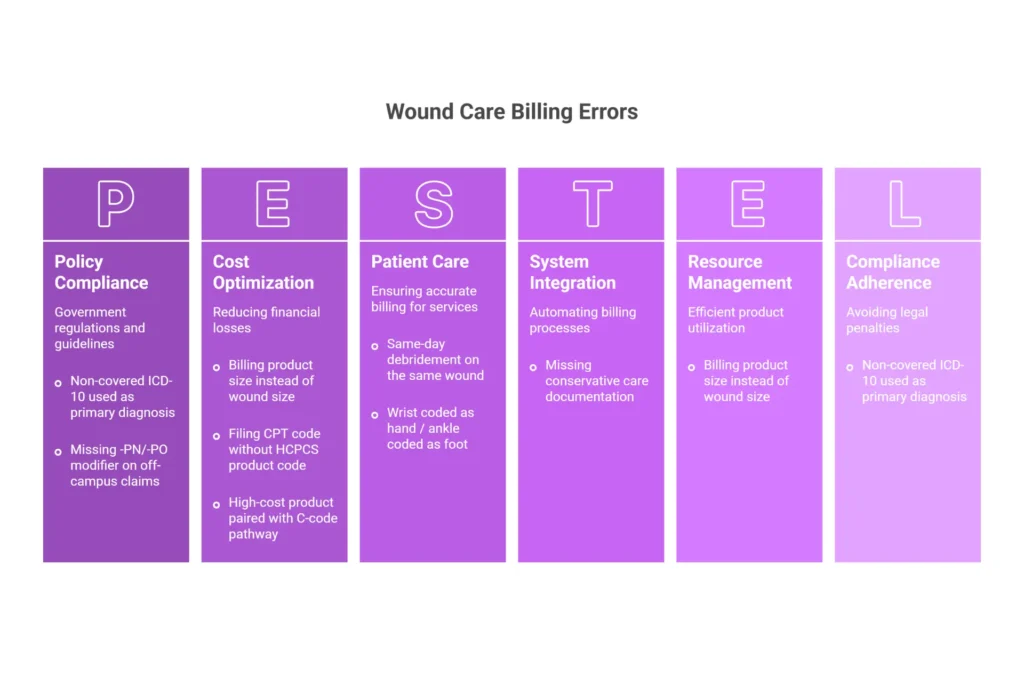
Top 8 Billing Errors and How to Stop Them in 2026
| Error | Why It Happens | Prevention Fix |
|---|---|---|
| Billing product size instead of wound size | Staff codes from the product package dimensions | Require pre-application wound measurement to be the billable unit |
| Filing CPT without HCPCS product code | Split-billing workflow | Claim scrubber rule: CPT 15271–15278 or C5271–C5278 must always trigger HCPCS product code on same claim |
| Same-day debridement on same wound | Misunderstanding bundling rules | Stage all debridement on prior conservative care visits |
| High-cost product paired with C-code pathway | Chargemaster not updated for 2026 cost-group assignments | Quarterly Addendum BB review with chargemaster sync every January, April, July, October |
| Wrist coded as hand / ankle coded as foot | Anatomy misunderstanding | Training: wrist = Group 1 (arm); ankle = Group 1 (leg) |
| Missing -PN/-PO modifier on off-campus claims | PBD status never verified | Build facility modifier into charge description master by location |
| Non-covered ICD-10 used as primary | Generic coding without LCD reference | LCD Group 1 covered diagnosis list built into pre-authorization workflow |
| Missing conservative care documentation | Weekly notes not collected before billing | Block charge entry until 4-week note packet is attached to encounter |
Stop Losing Revenue to
Preventable 2026 Denials
Skin substitute billing is one of the most denial-prone service lines in wound care — and 2026 added new layers of complexity. The WISeR pilot brought Medicare PA requirements to 6 states. The CY 2026 MPFS restructured non-facility product payment. New Q-codes entered the system. Practices that have not updated their workflows are paying for it in denial volume.
Prior Authorization for Skin Substitutes in 2026: WISeR Is Now Active
Prior authorization for skin substitutes is no longer a Medicare Advantage-only concern. The WISeR pilot went live January 1, 2026. If you practice in one of the six pilot states, Traditional Medicare Part B now requires pre-service PA for skin substitute procedures — the same as any Medicare Advantage plan.
PA Requirement by Payer Type — 2026 Status
| Payer | PA Required? | Trigger | Typical Timeline |
|---|---|---|---|
| Traditional Medicare Part B (WISeR pilot states: AZ, NJ, OH, OK, TX, WA) | Yes — active January 1, 2026 | All skin substitute procedure codes; pre-service submission required | 3–14 business days per CMS WISeR guidance |
| Traditional Medicare Part B (all other states) | No (post-pay audit model) | LCD compliance; audit risk is retrospective | N/A |
| Medicare Advantage (most plans) | Yes or formal utilization review | Product type, episode count, plan-specific rules | 3–14 business days typical |
| UHC Medicare Advantage (PCA-1-24-02596) | Post-service pre-payment review | Non-porcine products, all CTPs above frequency thresholds | 5–10 business days for review |
| Commercial / Employer Plans | Usually yes for high-cost grafts | Product cost > $500/application or non-formulary status | 3–14 business days |
| Medicaid / D-SNP | State and plan specific | High-cost CTPs; most managed-care plans require PA | Variable |
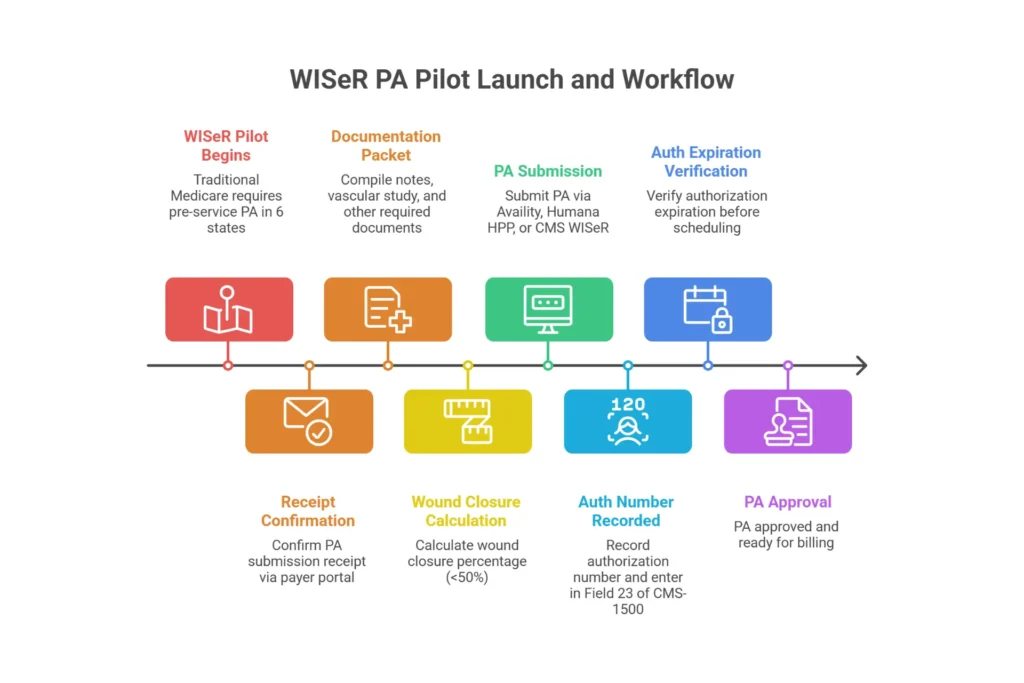
WISeR Pilot — What It Means for Your Practice Right Now
CMS launched the WISeR (Wound care and Skin substitute prior authorization) pilot effective January 1, 2026 in six states: Arizona, New Jersey, Ohio, Oklahoma, Texas, and Washington. Providers in these states must obtain pre-service prior authorization for skin substitute procedures under Traditional Medicare. Outside these six states, the post-pay audit model still applies for Medicare Part B FFS — but CMS has signaled interest in expanding WISeR based on pilot outcomes, which makes documentation compliance critical nationwide, not just in pilot states.
Monitor CMS’s Prior Authorization and Pre-Claim Review Initiatives page for expansion announcements throughout 2026.
Prior Authorization vs Pre-Determination — The Critical Distinction
A prior authorization is a binding coverage decision with an authorization number that protects the claim when entered in Field 23 of the CMS-1500. Without the authorization number on the claim, the authorization provides zero protection.
A pre-determination is advisory. It tells you what the payer is likely to cover. It does not guarantee payment and does not produce an authorization number. Never proceed with a costly graft based on a pre-determination alone.
8-Step PA Workflow
- Verify benefits and payer-specific PA rules during patient intake or the week before the scheduled graft date. In WISeR states, verify Medicare Part B PA requirements for the specific CPT codes you plan to bill.
- Confirm product formulary status — especially for UHC MA where non-porcine product preference is written into Policy PCA-1-24-02596.
- Compile the documentation packet: 4+ weeks of weekly wound notes with LxWxD measurements, <50% closure calculation, photos, vascular studies within 12 months.
- Calculate wound closure percentage: (Initial area − Final area) ÷ Initial area × 100. Example: Week 0 = 12 sq cm, Week 4 = 7.5 sq cm → 37.5% closure → qualifies (below 50%).
- Submit through the payer portal (Availity for UHC; Humana HPP for Humana; the CMS WISeR portal for Medicare in pilot states) with confirmation of receipt within 48 hours.
- Track submission status and follow up at day 3–5 for electronic submissions.
- Record the authorization number — enter it in Field 23 on every CMS-1500 claim for this service.
- Verify authorization expiration date before scheduling. Most PAs are valid 30–90 days. If the procedure is delayed, reapply.
PA Denial Appeals
First-level denial: submit a complete package including all 4 weeks of progress notes with measurements, closure percentage calculation, vascular study results, wound photos, fixation documentation, and product invoice. Request a peer-to-peer review with the plan’s medical director. Complete documentation packets succeed at approximately 65% on first-level appeal.
If first-level fails, escalate to an Independent Review Organization (IRO) for external review. Include LCD compliance documentation and relevant clinical literature supporting CTP efficacy for the patient’s wound type.
End-to-End Revenue Protection
for Every Skin Substitute
Your Practice Bills in 2026
GRAFT TRACK is a complete graft revenue management system — not billing software. It covers documentation workflow, 2026 code accuracy, pre-service PA for WISeR Medicare claims, Medicare Advantage PA submission, denial appeals, and audit-defense preparation. One program. Every claim. Zero gaps.
WISeR Medicare PA for AZ, NJ, OH, OK, TX, WA. MA plan PA for all plans. Auth number in Field 23. UHC PCA-1-24-02596 post-service review documentation. 8-step PA workflow end-to-end.
Voice Search Questions & Answers for Skin Substitute Billing
These questions reflect how wound care professionals ask about skin substitute billing through AI prompts, voice assistants, and smart search tools.
What CPT code do I use for a skin substitute applied to a diabetic foot ulcer? A diabetic foot ulcer on the foot falls in Group 2 (face, hands, feet, special sites). For wounds under 100 square centimeters, start with CPT 15275 for the first 25 sq cm, then add +15276 for each additional 25 sq cm or part thereof. Always pair with the HCPCS product code — for example, Q4186 for EpiFix — for the same wound area on the same claim. Verify 2026 OPPS or MPFS rates for your specific billing setting.
How many weeks of wound care notes do I need before billing a skin substitute to Medicare? Most LCD policies — including L35041, L36690, and L36377 — require a minimum of 4 consecutive weeks of documented standard wound care, with weekly clinical notes. Each note must include length × width × depth measurements and show that the wound healed less than 50% during that period. For Traditional Medicare claims in WISeR pilot states (AZ, NJ, OH, OK, TX, WA), this documentation must be compiled before submitting your pre-service prior authorization request.
Does Medicare now require prior authorization for skin substitute procedures? Yes — in six states. The WISeR pilot launched January 1, 2026 in Arizona, New Jersey, Ohio, Oklahoma, Texas, and Washington. Providers in those states must obtain pre-service prior authorization from Medicare before applying skin substitutes. In all other states, Traditional Medicare still uses a post-service audit model. Medicare Advantage plans require PA or utilization review nationwide regardless of WISeR status.
What happens if I use the wrong CPT code for a skin substitute claim? Wrong code selection creates either a procedure-device mismatch denial or an overpayment risk. Pairing a high-cost product (like Apligraf Q4101) with a low-cost C-code pathway (C5271) typically results in denial or severe underpayment — the practice absorbs the product cost. Pairing a low-cost product with the high-cost CPT pathway is considered overcoding and creates RAC audit exposure.
When do I use modifier -25 on a skin substitute claim? Modifier -25 belongs on the E/M code, not on the CPT procedure code. Use it when the evaluation and management visit on the same day involved genuinely significant clinical work beyond what is inherent in the skin substitute procedure — such as evaluating a new clinical problem or adjusting systemic treatment. Routine graft monitoring is not a separately billable E/M.
What is the difference between prior authorization and pre-determination for skin substitutes? A prior authorization produces a binding coverage decision with an authorization reference number that goes in Field 23 of your CMS-1500. A pre-determination is advisory only — it provides no authorization number and does not prevent denial. For WISeR pilot state Medicare claims and all Medicare Advantage claims, always obtain a binding prior authorization.
How do I calculate units for a skin substitute HCPCS product code? Bill the HCPCS product code in units equal to the square centimeters of wound area you treated — the recipient bed measurement, not the product size opened. For a 30 sq cm wound, enter 30 units in Box 24G. For the procedure code: 30 sq cm = 1 primary code unit (first 25 sq cm) + 1 add-on unit (remaining 5 sq cm rounds up to one additional 25 sq cm unit).
What documentation does a Medicare Advantage plan need for a skin substitute claim in 2026? Medicare Advantage plans require: 4+ weeks of weekly wound notes with measurements, wound closure calculation showing less than 50% healing, wound photographs, vascular assessment results within 12 months, no active infection documentation, and a completed prior authorization with the auth number in Field 23. UHC MA under Policy PCA-1-24-02596 additionally applies post-service, pre-payment review for non-porcine products.
Frequently Asked Questions: Skin Substitute Billing Codes 2026
How do you bill skin substitute codes?
Every skin substitute claim requires two codes on the same date of service: a CPT application code (15271–15278 for high-cost products, or C5271–C5278 for low-cost products in facility settings) and a HCPCS product code (Q-code or A-code specific to the product used). Select the CPT based on anatomical group (Group 1 for trunk/arms/legs; Group 2 for face/hands/feet/special sites) and total wound surface area. Always bill the wound recipient bed size — not the product sheet size — in units on the HCPCS product line.
What is the CPT code for skin substitutes?
CPT 15271 reports application of a skin substitute graft to the trunk, arms, or legs (Group 1 anatomical sites) when total wound surface area is under 100 sq cm. It covers the first 25 sq cm or less. The code includes graft fixation and simple wound bed preparation. Verify the 2026 APC 5054 rate in the current OPPS Addendum B for your CBSA.
What is CPT code 15275?
CPT 15275 reports skin substitute application to the face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, and multiple digits (Group 2) when total wound area is under 100 sq cm. Note: wrists are Group 1 (arm), and ankles are Group 1 (leg) — not Group 2.
What is the difference between 15275 and 15271?
The difference is anatomical group only. 15271 = trunk, arms, legs (Group 1). 15275 = face, scalp, neck, hands, feet, genitalia, multiple digits (Group 2). Both map to APC 5054 in the 2026 OPPS APC system. Select based strictly on wound location, not product type or procedure complexity.
Does CPT 15271 include debridement?
Simple wound bed preparation is included. Active surgical debridement (CPT 11042–11047) billed on the same wound on the same date is prohibited by NCCI edits (Indicator 0 — no modifier bypasses this). Perform all surgical debridement during the 4-week conservative care phase prior to graft application.
Does CPT 15271 need a modifier?
Not routinely. Modifiers are situation-specific: use -XS or -59 for multiple distinct wounds in different anatomical groups; use -25 on the E/M code when separately identifiable evaluation work was performed; use -PN or -PO for off-campus hospital outpatient department billing.
Can you bill 15004 and 15275 together?
Only with current NCCI edit verification. For the same wound on the same day, NCCI treats this pair as mutually inclusive. Check the live NCCI PTP edit file before billing this combination in any context.
Can you bill 15002 with 15271?
Same rule. For the same wound, same day, the NCCI edit treats surgical preparation as included in the application code. Review current NCCI edits for any separate-wound scenarios.
What are the four types of skin grafts?
(1) Split-thickness autograft — patient’s epidermis and partial dermis; (2) Full-thickness autograft — epidermis and complete dermis; (3) Composite graft — skin plus other tissue; (4) Skin substitutes/allografts — bioengineered, donor, or synthetic tissue-based products. Only skin substitutes require the paired CPT + HCPCS billing structure.
What are the four types of wound debridement?
(1) Surgical/sharp debridement (CPT 11042–11047); (2) Selective mechanical debridement (CPT 97597–97598); (3) Enzymatic debridement — topical collagenase agents; (4) Autolytic debridement — moisture-retentive dressings. Surgical debridement is the most defensible for LCD conservative-care documentation.
What is the ICD-10 code for skin substitute application?
There is no ICD-10-CM diagnosis code for “skin substitute application.” Medical necessity is established by the active wound diagnosis: E11.621 for Type 2 DFU, I83.011 for right venous leg ulcer, L89.153 for Stage 3 sacral pressure ulcer. Z48.816 may be used as a secondary code for follow-up visits.
What is the ICD-10 code for Z94.89?
Z94.89 means other transplanted organ and tissue status — used as a secondary diagnosis on follow-up encounters only. Never use it as the primary wound diagnosis.
What is the ICD-10 code for Z76.9?
Z76.9 is not an acceptable primary diagnosis for skin substitute claims. It triggers an MA130 medical necessity denial. Always use the specific wound etiology code as the primary diagnosis.
Does Medicare require prior authorization for skin substitutes in 2026?
Yes — in WISeR pilot states (AZ, NJ, OH, OK, TX, WA) effective January 1, 2026. Traditional Medicare Part B outside these six states still uses a post-pay audit model. Medicare Advantage plans require PA or formal utilization review nationwide.
What HCPCS code do I use for EpiFix?
Use Q4186 for EpiFix sheet form (MiMedx). This is the current standard code for the sheet product in 2026.
What HCPCS code do I use for Apligraf?
Use Q4101 for Apligraf (Organogenesis). High-cost group. Bill per square centimeter of wound recipient bed in Box 24G.
What is the HCPCS code for Dermagraft?
Use Q4106 for Dermagraft (Organogenesis). High-cost classification. Indicated for full-thickness DFUs.
What is the Q-code for AmnioFix?
Use Q4115 for AmnioFix (MiMedx). High-cost classification. For complete amniotic membrane billing guidance, visit our amniotic membrane graft billing guide.
Is Integra a high-cost skin substitute under Medicare in 2026?
Yes. Integra Bilayer Wound Matrix (Q4104) and Integra Omnigraft (Q4105) are classified as high-cost. These products pair with CPT 15271–15278. Verify the current 2026 Addendum BB for any quarter-specific changes.
Does Medicare Advantage require prior authorization for wound grafts in 2026?
Yes. Virtually all Medicare Advantage plans require either pre-service PA or post-service pre-payment review. UHC MA uses Policy PCA-1-24-02596. Blue Cross NC added PA requirements effective January 1, 2026. Always verify the current payer-specific policy before scheduling.
How often will Medicare pay for skin substitute applications?
Coverage is episode-based. Most LCDs and payer policies limit coverage to 4–8 applications per 12-week episode with documented wound improvement at each interval. A 5th or subsequent application requires additional clinical justification. Applications beyond frequency limits require thorough documentation and, for some payers, modifier -KX attestation.
What changed for skin substitute billing in 2026?
Three major changes: (1) The WISeR prior authorization pilot launched January 1, 2026 in six states, making pre-service PA mandatory for Traditional Medicare in AZ, NJ, OH, OK, TX, and WA. (2) CMS restructured the non-facility (physician office) payment model for skin substitute products under the CY 2026 MPFS Final Rule. (3) January 2026 HCPCS update cycle added new Q-codes — verify the full January 2026 release against your product formulary.
Conclusion: The Three Mistakes That Cost Practices the Most Money in 2026
Skin substitute billing has three failure points that generate the majority of losses, audits, and compliance exposure in wound care programs:
First: Filing the wrong application code for the product’s cost group — high-cost product with a low-cost C-code, or low-cost product with a high-cost CPT. This single error can generate either denied claims or overpayments flagged during RAC review.
Second: Incomplete conservative care documentation. Four consecutive weekly notes with measurements showing less than 50% wound closure is not a suggestion. It is the gatekeeper requirement for Medicare and Medicare Advantage coverage. In WISeR states, it is also the foundation of your pre-service PA submission. Every claim without it is vulnerable.
Third: Missing or incorrect modifiers for off-campus billing, multiple wounds, or same-day E/M services. In 2026, add the WISeR PA requirement to this list — a valid PA that is not entered in Field 23 protects nothing.
The fix for all three is a built-in workflow, not a better cheat sheet. At EliteMedFinancials.com, our wound care billing and RCM team builds those workflows for practices. We also offer end-to-end revenue cycle management through our RCM services, dedicated medical coding oversight, and the GRAFT TRACK Verified Graft Program — a complete system for documentation, prior authorization, product code management, and audit defense that is fully updated for 2026.
Ready to stop leaving money on the table?
→ Schedule a Free Wound Care Billing Audit
→ Explore the GRAFT TRACK Program
References: CMS Quarterly Addenda Updates — cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient-pps/quarterly-addenda-updates | AMA CPT Code Set — ama-assn.org/practice-management/cpt/cpt-overview-and-code-approval | CMS WISeR Prior Authorization Pilot — cms.gov/data-research/monitoring-programs/medicare-fee-service-compliance-programs/prior-authorization-and-pre-claim-review-initiatives

