
Wound care graft prior authorization is required by most commercial payers, Medicare Advantage plans, and Dual Special Needs Plans (D-SNPs) before the first application of any skin substitute, cellular tissue-based product (CTP), or amniotic membrane product. Without a valid prior authorization number documented in Field 23 of the CMS-1500, your claim will be denied — period — regardless of how solid the clinical documentation is. This guide gives you the exact workflow, documentation checklist, payer-specific rules, and denial prevention strategies that wound care providers use to achieve first-pass approval rates above 90% in 2026.
Table of Contents
What Wound Care Graft Prior Authorization Means — and Why It Delays Treatment
Let me start with the honest version of this conversation — the one most billing articles skip. Prior authorization for wound care grafts is not a formality. It is not something you can backfill after a denial. And it is not something your clinical staff can manage alongside everything else they already do without a system.
Prior authorization (PA) is a payer’s right to review and approve — or refuse — a proposed treatment before it is delivered. In the context of wound grafts, it means that before you apply an amniotic membrane product like AmnioRepair (Q4235), EpiFix (Q4131), or Apligraf (Q4101) to a chronic wound, many payers need to confirm that the product selection, the wound type, and the documented treatment history all meet their specific coverage criteria. They want to see the evidence before they commit to paying for a product that can cost anywhere from $800 to $4,000 per application.
This is different from how traditional wound dressings work. Nobody at UnitedHealthcare is reviewing your order for a foam dressing. But skin substitutes and CTPs sit in a completely different clinical and financial category — one that has been under escalating payer scrutiny since CMS’s post-pay audit findings revealed utilization patterns that didn’t meet coverage criteria in several Medicare Advantage markets.
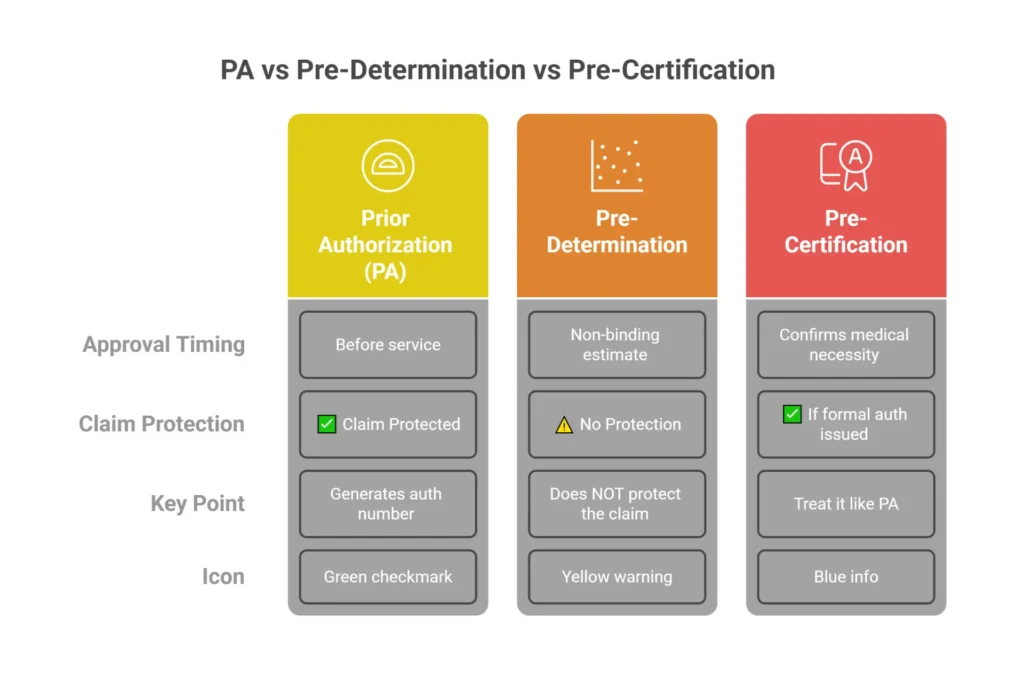
Prior Authorization vs. Pre-Determination vs. Pre-Certification — What’s the Difference?
These three terms are used interchangeably in the wild and they are not the same thing. Confusing them costs practices real money.
| Term | What It Means | Wound Graft Context |
|---|---|---|
| Prior Authorization (PA) | Payer reviews and approves the service BEFORE delivery | Required for CTPs and amniotic products by most MA and commercial plans |
| Pre-Determination | Non-binding estimate of likely coverage | Useful for planning; does NOT protect the claim from denial |
| Pre-Certification | Confirms medical necessity criteria are met | Sometimes used interchangeably with PA by MA plans — treat it like PA |
| Referral | PCP direction to a specialist | Separate from PA; some plans require both simultaneously |
The critical distinction: a pre-determination letter does not protect your claim. Providers sometimes call ahead to ask “will this be covered?” and receive a verbal or written confirmation that looks like an approval. It is not. Only a formal prior authorization with a trackable authorization number that goes into CMS-1500 Field 23 protects the claim.
The AmnioRepair Coding Reference Guide (Zimmer Biomet) states it directly: “Prior authorization/pre-determination is recommended prior to administration of a skin substitute graft. The payer will want to review the specific product proposed for use, dosage, and medical necessity.” That framing — “recommended” — understates the reality. For most Medicare Advantage and commercial payers, it is required.
Why Wound Grafts Specifically Trigger Prior Authorization
There are two forces driving this. The first is product cost. Advanced wound care products — particularly amniotic membrane grafts and cellular tissue-based products — are among the most expensive items billed under outpatient wound care CPT codes. A single 4×4 cm amniotic membrane application can carry a wholesale acquisition cost of $1,500 to $3,500 depending on the product. When claims are being filed for multiple applications per patient at those price points, payers pay attention.
The second force is utilization history. United Healthcare’s Medicare Advantage division made this explicit in their Clinical FAQ (Policy PCA-1-24-02596, issued August 28, 2024): the policy was created because UHC’s MA plans discovered skin substitute utilization patterns that did not appear to meet coverage criteria. That’s not speculation — that’s a payer telling you in writing why they built an enhanced review layer on top of standard LCD criteria. Their response was to implement a post-service, pre-pay clinical review model for non-porcine skin substitutes across multiple MA plan types.
CMS responded to broader utilization concerns nationally by reclassifying skin substitute products in 2025–2026 OPPS rulemaking, creating high-cost and low-cost payment categories that changed the reimbursement math for providers. The coverage scrutiny followed the reimbursement change, not the other way around.
The Real Cost of a Missed Prior Authorization
Here’s the number that makes wound care practice managers wake up at night: a single missed prior authorization on a 4×4 cm amniotic product means a potential write-off of $2,000 to $8,000 depending on the product and payer. There is no retroactive PA option. Once the graft has been applied, the window for pre-service authorization has passed.
The downstream consequences compound quickly. If you didn’t have a valid Advance Beneficiary Notice (ABN) in place before applying the graft to a Medicare patient, the patient cannot be billed either. The practice absorbs the full product cost plus the application fee with no recovery path. And the staff time required to identify, rework, and attempt recovery on a retroactive denial runs 2–4 hours per claim on average — hours that come at the direct expense of new authorizations being submitted.
Real-world example: A wound care practice applying 3 amniotic grafts per week without a structured PA tracking system will miss roughly 1–2 authorizations per month through normal administrative gaps — scheduling mix-ups, insurance changes between visits, product substitutions, or staff turnover. At an average write-off of $3,500 per missed PA, that’s $3,500 to $7,000 in preventable monthly revenue loss. Annualized, that’s $42,000–$84,000 walking out the door.
For more on how Medicare coverage rules interact with your billing requirements, see our complete Medicare wound care graft coverage guide.
Which Wound Grafts and Amniotic Products Usually Trigger Prior Authorization in 2026
Not every wound care product triggers the same level of scrutiny. Knowing which products face the most friction — and why — lets you build your authorization workflow to match the risk level of each product you stock.
Skin Substitute and CTP Products That Almost Always Require Prior Auth
The following table is based on payer policy analysis, the CMS HCPCS coding structure, and product-specific LCD criteria. This is not exhaustive — check individual payer policies at benefit verification — but it gives you the baseline risk profile for common products.
| Product | HCPCS / Q-Code | Category | PA Required? | Key Notes |
|---|---|---|---|---|
| Apligraf | Q4101 | Cellular (living bilayer) | Yes — most payers | One of the most scrutinized products due to cost and volume |
| Dermagraft | Q4106 | Cellular (human fibroblast-derived) | Yes — most payers | Requires DFU indication documentation |
| EpiFix | Q4131 | Amniotic (dehydrated) | Yes — MA and commercial | MIMEDX product; UHC MA applies enhanced review |
| AmnioFix | Q4115 | Amniotic (injectable form) | Yes — MA and commercial | MIMEDX injectable; wound type criteria apply |
| AmnioRepair | Q4235 | Amniotic (lyophilized) | Yes — most commercial | Zimmer Biomet; UPC in Field 19; Q4235 + JC billing |
| PuraPly AM | Q4174 | Acellular (collagen/ORC) | Yes — MA and commercial | Organogenesis; product-specific PA requirements by plan |
| Kerecis Omega3 | Q4204 | Xenograft (fish skin) | Yes — most payers | Emerging category; some plans still classifying coverage |
| Grafix | — | Cryopreserved placental | Yes — all major payers | Among the highest-cost products; strict clinical criteria |
| Oasis Wound Matrix | Q4102 | Acellular (porcine) | Varies — often lower scrutiny | Porcine-derived; see note below on UHC MA policy |
| Biasyn | Q4161 | Amniotic | Yes — MA plans | Confirm formulary status per plan |
The porcine exception worth knowing: UHC Medicare Advantage’s enhanced post-pay review policy specifically targets non-porcine skin substitutes. Porcine-derived products like Oasis face a lighter review burden under this policy. This matters when you’re selecting a product for a Medicare Advantage patient — the authorization friction is genuinely lower for porcine-derived acellular dermal matrices at certain MA plans. That doesn’t mean porcine products are always the better clinical choice, but it is a real administrative consideration worth discussing with your treating physician.
Amniotic Membrane Grafts — Wound vs. Ocular Prior Authorization
This is an area where practices create expensive errors because they assume the same PA pathway applies across all amniotic membrane products. It doesn’t.
Wound application (CPT 15271–15278 + Q-code): Governed by LCD wound criteria. The fundamental requirements are chronic wound status (typically 4+ weeks of non-healing), wound type confirmation (DFU, VLU, pressure injury), vascular assessment on file, and documented failure of at least four weeks of conservative treatment. This is the PA pathway that the main body of this guide covers.
Ocular application (CPT 65778 — placement without sutures): Completely separate authorization pathway. CPT 65778 is governed by ophthalmic LCD criteria, not wound LCDs. Documentation templates designed for wound PA requests will not satisfy an ocular PA reviewer. This is an entirely different clinical indication set.
For ocular amniotic membrane applications, the Oklahoma Health Care Authority (OHCA) guideline (effective March 17, 2025) provides a clear framework that reflects payer standards nationally. Covered indications under ocular amniotic membrane criteria include:
- Neurotrophic keratitis that has not responded to conservative therapy
- Corneal ulcers or melts not responding to initial management
- Corneal perforation with active inflammation following transplant
- Bullous keratopathy — palliative indication only, for non-surgical candidates
- Partial limbal stem cell deficiency
- Moderate or severe Stevens-Johnson syndrome
- Persistent epithelial defects not responding within 2 days of initial treatment
- Severe dry eye meeting DEWS grade 3–4 criteria after completion of management algorithm Steps 1–3
- Moderate to severe acute ocular chemical burns
Billing note: Never submit a wound application documentation template for an ocular amniotic membrane prior auth request. Reviewers will reject it immediately, and the delay adds 5–10 business days to the authorization timeline. For a detailed look at amniotic membrane billing across wound and ocular categories, see our guide to amniotic membrane graft billing CPT codes and Q codes.
Products Under Emerging Coverage Development
A few product categories are in active coverage policy development in 2026 and deserve monitoring: fish skin xenografts (Kerecis category), stromal vascular fraction-derived products, and some exosome-based wound applications. If your practice is evaluating any newer CTP or amniotic product not on a published LCD, conduct a thorough pre-authorization verification before the first application — treating these like a commercial insurance product with no established coverage precedent.
When Wound Graft Prior Authorization Is Required by Payer Type
The question of when prior authorization is required is payer-specific, plan-specific, and in some cases, state-specific. The same patient presenting with the same wound type can require entirely different authorization steps depending on whether they carry traditional Medicare, a UHC Medicare Advantage plan, an Aetna commercial plan, or a D-SNP. Here’s how to navigate each one.
Traditional Medicare (Part B) — No Pre-Service PA, But Post-Pay Risk Is Real
Let’s clear up the misconception that gets practices into trouble: original Medicare Part B does not require pre-service prior authorization for most wound graft procedures. You can apply a covered skin substitute to a qualifying wound without submitting an authorization request first.
What Medicare does instead is conduct post-payment audits — through Medicare Administrative Contractors (MACs), Recovery Audit Contractors (RACs), and Unified Program Integrity Contractors (UPICs) — that function as a retroactive authorization review. If your medical record doesn’t support the LCD criteria that were in effect at the time of service, the claim gets denied on audit and the payment gets recouped. The effect on your bank account is identical to a failed pre-service PA. The difference is the timeline — post-pay audits can arrive 12 to 36 months after the service date.
The applicable LCDs vary by MAC jurisdiction. The three most active in wound graft coverage as of 2026:
- CGS Administrators (J15): LCD L36690 — applicable in Kentucky and Ohio
- Novitas Solutions (JH/JL): LCD L35041 — applicable in Arkansas, Colorado, DC, Delaware, Louisiana, Maryland, Mississippi, New Jersey, New Mexico, Oklahoma, Pennsylvania, and Texas
- First Coast Service Options (JN): LCD L36377 — applicable in Florida, Puerto Rico, and U.S. Virgin Islands
If your state isn’t covered by an active LCD for CTPs, your MAC applies broader medical necessity criteria — which paradoxically increases your audit exposure because there’s no defined criteria checklist to work from.
Medicare Advantage Plans — Prior Authorization Is Standard
Medicare Advantage plans are a different world entirely. These plans have the legal authority under CMS to impose prior authorization requirements that don’t exist in original Medicare. Most do. The level of pre-service review, the documentation requirements, and the turnaround times vary significantly from plan to plan — but the baseline expectation for wound graft applications is that authorization is required before service.
The most detailed example of how MA PA policies work in practice comes from UnitedHealthcare’s Medicare Advantage Clinical FAQ (Policy PCA-1-24-02596, effective August 15, 2024). UHC uses a post-service, pre-pay clinical review model for non-porcine skin substitutes — meaning they don’t require a formal pre-service authorization, but they hold the claim for clinical review after submission. If documentation is requested and not provided, the claim is denied.
UHC Medicare Advantage plans covered under this policy:
- UHC Medicare Advantage Individual (including UHC West)
- UHC Medicare Advantage Group — EGWP and United Retiree Solutions
- UHC Medicare Advantage + Community Plan D-SNPs, including Optum at Home
- Institutional Special Needs Plans (I-SNPs)
UHC plans NOT covered under this specific policy:
- Institutional-Equivalent SNP (IE-SNP)
- Preferred Care Network / Preferred Care Partners
- Peoples Health plans
- Rocky Mountain Health Plans
This distinction matters for your verification workflow. Don’t assume all UHC plans operate the same way — the four excluded plan types above don’t trigger UHC’s enhanced skin substitute review, which means your documentation and authorization approach for those patients differs.

| Plan Type | PA Required? | Review Type | Notes |
|---|---|---|---|
| MA Individual (UHC) | Yes | Post-service, pre-pay clinical review | Determinations via UHC Provider Portal |
| MA Group / EGWP (UHC) | Yes | Post-service, pre-pay | Same policy applies |
| D-SNP / Community (UHC + Optum at Home) | Yes | Post-service, pre-pay | Includes Optum at Home patients |
| I-SNP | Yes | Post-service, pre-pay | Institutional SNP — same UHC policy |
| IE-SNP | No | Excluded from this policy | Separate verification required |
| Peoples Health / Rocky Mtn / Preferred Care | No | Excluded from this policy | Verify per plan policy |
| Non-UHC MA Plans | Varies | Typically pre-service | Plan-specific PA required |
For non-UHC Medicare Advantage plans: Humana, Aetna MA, Cigna MA, and BCBS MA affiliates all require pre-service prior authorization for skin substitutes. The specific form, turnaround time, and portal vary — but the authorization requirement itself is consistent.
Commercial Payers (Non-Medicare)
Commercial plan requirements vary widely by carrier and state, but the general direction is clear: skin substitutes and CTPs are flagged for pre-service authorization at most large commercial carriers.
- Aetna: Pre-service PA required for CTPs exceeding $500 in acquisition cost. Product-specific criteria apply.
- Blue Cross Blue Shield: State-level medical policy governs; most BCBS affiliates require PA for non-porcine products. Review the specific affiliate’s wound care policy.
- Cigna: PA required for the “advanced wound care” product category, which includes amniotic and cellular products.
- WellCare (now Centene): PA required per MA medical policy for skin substitute applications.
- Tricare: Pre-authorization required for skin substitute procedures. Submit through the Humana Military provider portal.
Medicaid and D-SNP Patients — Double Verification Required
Patients enrolled in Dual Special Needs Plans carry both Medicare and Medicaid benefits. The coordination rules are complex, and in practice this means your authorization workflow needs to address both payers. Many D-SNP plans use MA prior authorization rules for their Medicare-covered services, but some state Medicaid programs impose additional requirements.
The ProCare Advantage Wound Care Prior Authorization Form (V3.10.2025) is a representative example of what a MA/D-SNP prior authorization form looks like in practice. It requires wound measurements (initial and current), drainage type and volume, wound thickness classification, full treatment history, and CPT/HCPCS code assignment upfront — before your request even gets to a clinical reviewer.
For patients with both Medicare and Medicaid coverage, always verify both payers during the initial insurance verification step. Missing the Medicaid component can create secondary billing complications even when your Medicare authorization is clean.
Exact Documentation Needed for Wound Graft Insurance Approval
This section is where authorization requests succeed or fail. The documentation package is not something to assemble after the payer requests it — by that point, you’re already in a reactive position, working against a deadline, and at risk of an automatic denial if you miss it.
The framework below organizes required documentation into two categories: medical necessity documentation (patient-side) and product documentation (product-side). Both must be complete before submission.
Category 1: Medical Necessity Documentation (Patient-Side)
1. Wound type and ICD-10 code
- E11.621 — Type 2 diabetes mellitus with foot ulcer (most common DFU code)
- E10.621 — Type 1 diabetes mellitus with foot ulcer
- I83.009 — Varicose veins, unspecified lower extremity, with ulcer
- L89.xxx — Pressure injury (site and stage-specific)
- L97.xxx — Non-pressure chronic ulcer of lower extremity
The ICD-10 code must match a wound type on the applicable LCD covered indications list. A mismatch between what’s documented clinically and what’s coded is one of the most common PA denial triggers.
2. Wound duration and chronicity Document the wound’s origin date — either from the initial treating provider’s records or the patient’s report with reasonable medical certainty. The wound must meet chronicity criteria: most LCD policies define “chronic” as 4+ weeks of non-healing despite appropriate conservative treatment. This is not implied — it must be explicitly stated in the medical record.
3. Wound measurements — initial AND current
- Length × width × depth in centimeters
- Total wound area in square centimeters (L × W = area used for CPT unit calculation)
- Tunneling depth and direction if present
- Undermining measurement if present
Both the initial measurement (at wound onset or first visit to your practice) and the current measurement at time of graft application are required. Without comparative measurements, you cannot demonstrate treatment failure — and treatment failure is the clinical justification for escalating to a graft.
4. Drainage documentation None / Scant / Minimal / Moderate / Heavy — Copious. Document this at every wound care visit. Payers use drainage patterns to assess wound acuity and the appropriateness of an advanced wound care product.
5. Conservative treatment history — 4 consecutive weeks minimum This is the requirement that trips up the most practices. You need documentation from at least four consecutive weeks of wound care that demonstrates:
- The wound was treated with appropriate standard therapies (moisture-balanced dressings, debridement, offloading for DFUs, compression therapy for VLUs)
- The wound failed to progress toward closure (less than 50% area reduction over the 4-week period is the common clinical threshold)
- Each visit is documented with date, provider, treatment applied, and wound measurements
Gaps in visit documentation are treated by reviewers as absence of treatment. If a patient missed a week, document the missed appointment and the reason. A partial record is worse than a complete record with documented gaps.
6. Vascular assessment For diabetic foot ulcers: an ankle-brachial index (ABI) or Doppler study must be on file, typically within 12 months of the graft application. Some plans require a more recent vascular assessment. Document the result, the date, the testing provider, and any clinical interpretation.
7. Infection management documentation If the wound was infected at any point, document the clinical findings, the treatment applied, and the resolution. An actively infected wound at the time of graft application is a clinical exclusion criterion under most LCDs — document clearly that infection was controlled before graft application.
Category 2: Product Documentation (Product-Side)
8. Product identification
- Product name (e.g., AmnioRepair)
- HCPCS/Q-code (e.g., Q4235)
- NDC or UPC code (from product packaging)
- WAC (wholesale acquisition cost) or actual invoice price
Per the AmnioRepair Coding Guide, Field 19 of the CMS-1500 must contain: product name, NDC/UPC/HRIC code, and invoice price. This is how the payer verifies the specific product and cost. Missing any of these creates a claim edit that results in a hold or denial.
9. Product size and quantity
- Graft size in square centimeters
- Number of units (= number of sq cm applied, not number of packages)
- Lot number from packaging
10. Clinical rationale for product selection For high-cost products and MA plans: document why this specific product was selected. Reference published clinical evidence for the product. For amniotic membrane products specifically, documentation should address why conservative care has failed and what clinical evidence supports amniotic membrane application for this wound type.
11. CMS-1500 Field 23 — Authorization number If your plan requires pre-service PA, the authorization number goes in Field 23 before the claim is submitted. If UHC MA is using post-service review, you document the pre-service verification steps and leave the field for the determination number once received. Either way, this field is your proof of authorization — never submit a skin substitute claim without it populated.
The Complete Pre-Submission Documentation Checklist
Documentation Checklist
All 13 items must be verified prior to submission. Incomplete documentation is the leading cause of wound graft PA denials.
For deeper context on how these documentation requirements connect to Medicare’s coverage standards and audit exposure, our wound care billing guidelines covers the complete compliance framework.
Step-by-Step Workflow for Getting Wound Graft Prior Authorization Approved
Here is the authorization workflow used by high-performing wound care practices — the ones that run approval rates above 90% and rarely see denials for documentation deficiencies. Eight steps, sequenced correctly, with realistic time estimates for each so you can staff appropriately.
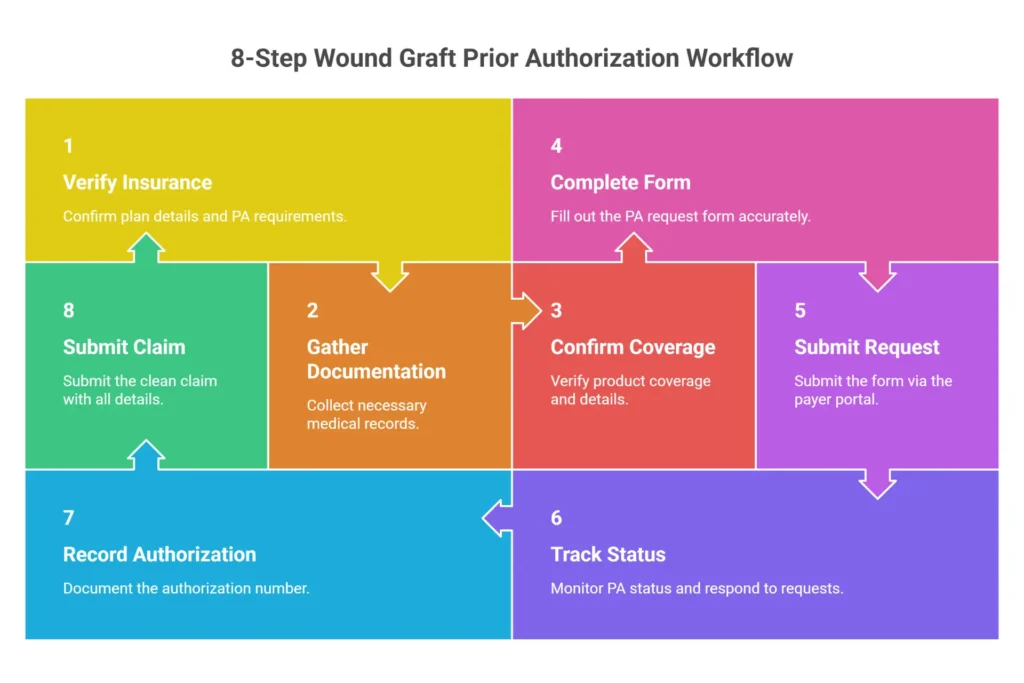
Step 1 — Patient Insurance Verification (Day 0, Before Any Appointment)
Time: 15–30 minutes
The authorization workflow begins before the patient walks in the door. During intake, verify:
- Plan type: Original Medicare vs. Medicare Advantage vs. Commercial vs. Medicaid/D-SNP
- Whether the specific product being considered is on the payer’s approved formulary
- Whether PA is required for wound graft / skin substitute procedures
- Payer’s PA submission channel: portal, fax, or phone
- Whether the plan uses pre-service or post-service review (critical distinction for UHC MA patients)
For UHC Medicare Advantage plans using the post-service review model: the clinical verification steps still happen before the graft is applied. You are documenting pre-service compliance even though the formal claim review happens after. This is not a loophole — it’s the model UHC designed. The documentation of your pre-application verification is what protects you if UHC requests records after claim submission.
Step 2 — Gather Medical Necessity Documentation (Days 0–7)
Time: 30–60 minutes
Pull the complete documentation package before the PA request is submitted. This means:
- Wound care visit notes from at least the prior four weeks
- Wound measurements from every visit — initial and most recent
- Vascular assessment (ABI/Doppler) — confirm it’s within 12 months
- Record of conservative care applied — specific dressings, debridement method, offloading used
- Infection treatment documentation if applicable
- Confirmed ICD-10 code assignment before the form is completed
Do not submit a PA request and then locate the supporting documentation reactively if the payer asks. By the time they ask, you may have 5 business days to respond — and assembling clinical records under that deadline is how good claims become preventable denials.
Step 3 — Select Product and Confirm Its Coverage Status
Time: 10–20 minutes
Not all products are created equal in the payer’s eyes. Before finalizing product selection:
- Confirm the Q-code/HCPCS code is on the payer’s covered product list
- For MA plans: confirm the product is not excluded by the plan’s specific formulary
- Identify whether the product is classified as high-cost or low-cost under OPPS rules
- For UHC MA patients: confirm whether the product is porcine or non-porcine — non-porcine products face UHC’s enhanced review
- Confirm lot number, product size, and invoice price for CMS-1500 Field 19
This is also the step where product substitution decisions should happen — if the clinically preferred product faces significant PA friction for this specific payer, this is the conversation to have before ordering, not after.
Step 4 — Complete the Prior Authorization Request Form
Time: 20–40 minutes
Use the payer’s specific form. For ProCare Advantage patients, the current form (V3.10.2025) requires:
- Patient information, plan ID, provider NPI
- Wound type, ICD-10 code, wound duration
- Initial measurements (date + dimensions) and current measurements (date + dimensions)
- Drainage type and volume
- Wound thickness: Superficial / Partial thickness / Full thickness / Deep
- Treatment history — conservative care used with dates
- CPT and HCPCS codes, product name, Q-code
- Request type: Initial vs. Extension of services
- Priority: Routine vs. Urgent/Serious jeopardy
ProCare Advantage submissions go to fax 1-833-610-2399 or via provider portal at secure.healthx.com/ProCareAdvantage.Provider.
Every field matters. Incomplete forms are returned, which adds 3–7 business days to your authorization timeline while you resubmit. Make it a rule: no PA form goes out with a blank field.
Step 5 — Submit via Preferred Payer Channel
Time: 5–10 minutes
Portal submission is always the preferred method — it creates a timestamp and generates a reference number that can be tracked. Fax is acceptable but requires a confirmation receipt with timestamp kept on file. Phone submission is a last resort only — if you do it, document the agent name, reference number, date, time, and call duration immediately after hanging up.
For UHC Medicare Advantage: submit through the UnitedHealthcare Provider Portal. Determinations are sent back through the portal. Monitor the portal — UHC does not always send proactive notifications when documentation is requested.
Step 6 — Track Status and Follow Up
Time: 10–15 minutes at days 3 and 5
Build a PA tracking log. Every active authorization request needs a row that includes: patient name, date of birth, payer, product requested, submission date, tracking/reference number, determination deadline, follow-up dates, and final status.
Standard turnaround times to expect:
- Routine PA: 3–5 business days
- Urgent / Serious jeopardy: 24–72 hours
- UHC MA post-service review: Variable — typically 10–30 days after claim submission
If the payer requests additional clinical documentation, respond within their stated deadline — commonly 5–10 business days. Missing this response window results in automatic denial for “lack of information,” and these denials are almost always preventable.
Step 7 — Receive Determination and Document the Authorization Number
Time: 5 minutes
When approval comes through:
- Record the authorization number in CMS-1500 Field 23 immediately
- Confirm the authorization covers the specific product, the number of approved applications, and the valid start/end dates
- Confirm the authorization is valid for the actual planned service date — authorizations expire
If the determination is a partial approval (for example, 2 of 4 requested applications approved), document the approval clearly and bill only the covered applications. Submit an appeal for the remaining 2 applications with supplemental documentation. Do not bill beyond the authorized quantity.
Step 8 — Apply Graft and Submit Clean Claim
Time: N/A (clinical) + 20 minutes (billing)
Once authorization is confirmed and the clinical visit is complete:
- Bill CPT 15271–15278 based on wound size, location, and graft type
- Include the Q-code with JC modifier (the graft was used)
- Enter the authorization number in Field 23
- Enter product name, NDC/UPC, and invoice price in Field 19
- Bill wastage separately: unused portion on a separate line with JW modifier; if no waste, add JZ modifier
- Units in Field 24G = actual square centimeters used (a 4×4 cm graft = 16 units)
Getting wound care graft claims right at every step — from billing CPT codes to managing the prior auth — is the kind of work our wound care billing and coding services team handles daily. Clean claims start before the patient walks in, not when the encounter is over.
| Payer Type | Routine PA Turnaround | Urgent/Expedited | Post-Service Review |
|---|---|---|---|
| Original Medicare | No pre-service PA | N/A | Post-pay MAC/RAC audit |
| UHC Medicare Advantage | Post-service review | N/A | Pre-pay hold after claim submission |
| Other MA Plans | 3–5 business days | 24–72 hours | Varies by plan |
| Commercial (Aetna, BCBS, Cigna) | 3–7 business days | 24–72 hours | Not standard |
| Medicaid / D-SNP | 3–14 days | 24–72 hours | State-specific |
| ProCare Advantage MA | Routine or urgent per form | Per clinical need | Per MA regulations |
Top Reasons Wound Graft Prior Auth Gets Denied — and How to Prevent Each One
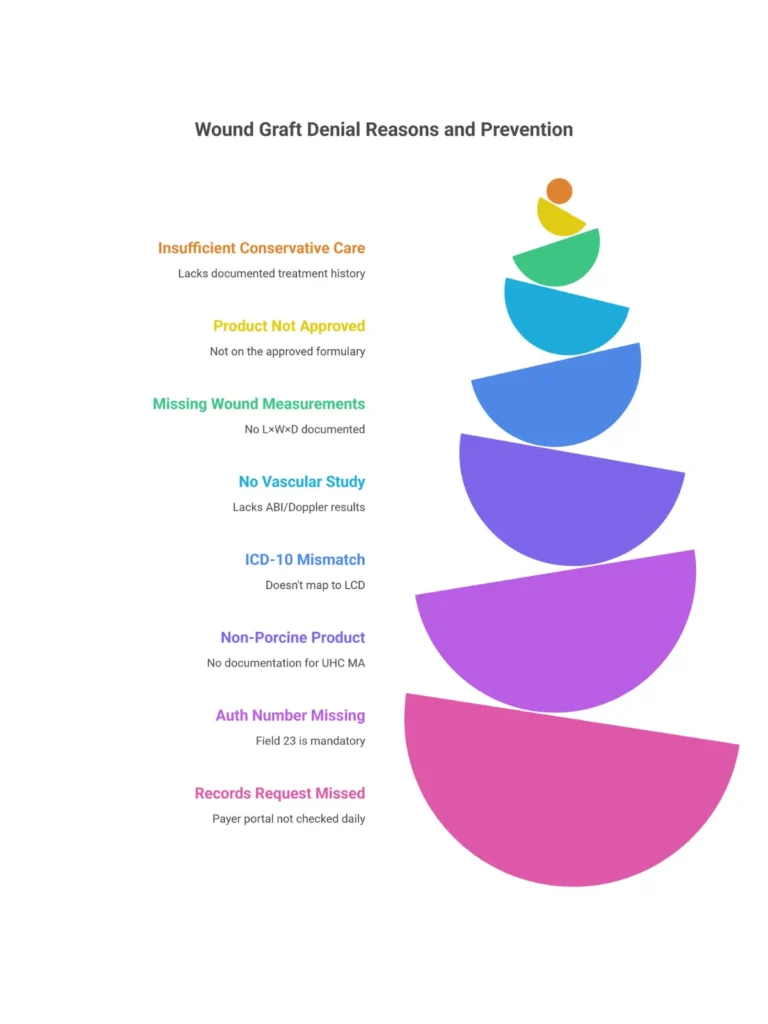
Denial patterns in wound graft prior authorization are highly predictable. After working through hundreds of authorization requests and appeals, the same eight reasons account for the vast majority of denied requests. Every one of them is preventable.
| Denial Reason | Root Cause | Prevention Fix |
|---|---|---|
| Insufficient conservative care documentation | Fewer than 4 weeks documented, or visit notes missing for some weeks | Document every visit; 4+ consecutive weeks minimum with dates and treatments specified |
| Product not on approved formulary | Q-code/product excluded from plan’s covered list | Run product formulary check during IVR before ordering the product |
| Missing wound measurements | No initial or current measurements in the record | Measure at every visit: L × W × D, cm², tunneling, undermining |
| No vascular study on file | ABI/Doppler missing or older than 12 months | Verify and update vascular study before graft application |
| ICD-10 mismatch | Code doesn’t map to a covered wound type on the LCD | Confirm ICD-10 matches covered indications before submitting |
| Non-porcine product on UHC MA without documentation | Selected without verifying UHC MA’s enhanced review criteria | Run formulary check for every MA patient; document clinical rationale for product selection |
| PA number missing from CMS-1500 Field 23 | Auth obtained but not entered before billing | Make Field 23 a mandatory billing checkpoint — no PA number = claim hold |
| Records request missed | Payer requested clinical documentation; practice didn’t respond in time | Build a PA status tracker; check portal daily; respond within 5 business days of any records request |
What Happens When UHC Medicare Advantage Denies a Wound Graft Claim
UHC’s Medicare Advantage post-service review process deserves special attention because it’s structured differently than traditional PA. Under this model, you don’t submit a pre-service PA request — you submit the claim, and UHC may then pull it for clinical review before payment.
When that happens, you’ll receive a notification through the UHC Provider Portal requesting clinical documentation. The clock starts ticking from that request date. If records aren’t provided in time, the claim is denied for lack of information — not for clinical inappropriateness. That’s a different denial reason with a different appeal path.
UHC sends the determination to the provider via the Provider Portal. Members receive notification by mail. To resolve a denial or pursue an appeal, follow the instructions in the denial correspondence exactly.
UHC Provider Contact for appeals and status:
- Providers contracted with UHC: 888-676-7768
- Providers contracted with Optum: 800-873-4575
How to Appeal a Denied Wound Graft Prior Authorization
A denied wound graft prior authorization is not the end of the road. Most initial denials are recoverable with a properly structured appeal. Here’s the framework:
Level 1 — Internal Reconsideration: File with the payer within 30 days of the denial date. Include any additional documentation that addresses the specific denial reason. Attach a physician-signed letter of medical necessity that references the applicable LCD criteria and explicitly documents the clinical evidence supporting the product. Generic appeal letters fail — the letter must address the specific reason stated in the denial.
Level 2 — Independent Review: If the first-level appeal is denied, escalate to an independent review organization (IRO). Provide the complete clinical record, LCD citations, and published peer-reviewed evidence supporting the product’s efficacy for this wound type. For amniotic membrane products, there is a growing body of published clinical trial data that supports medical necessity — use it.
Level 3 (MA Plans) — Medicare Appeals Process: Medicare Advantage plans must process appeals under Medicare rules. This means you have access to the full five-level Medicare appeals process — internal reconsideration, IRO, Administrative Law Judge hearing (for amounts ≥$180), Medicare Appeals Council, and Federal District Court. This is not theoretical — wound graft claims have been successfully recovered at the ALJ level when the documentation was complete and the clinical rationale was strong.
Appeal success rates improve significantly when you: address the specific denial reason rather than resubmitting the same package, include wound photographs if available, have the treating physician sign a new LMN specifically addressing the denial, and cite LCD criteria by number with the specific language met.
Our denial management and AR services team specializes in wound graft appeal management — if you’re seeing recurring denial patterns on graft claims, that’s a solvable problem.
🧮 Wound Graft Prior Authorization Revenue Risk Calculator
See how much revenue your practice is risking from missed or unmanaged prior authorizations — and what recovery looks like.
Estimates based on industry averages and your inputs. Actual results vary. Contact EliteMedFinancials for a practice-specific revenue analysis.
CMS, Medicare Advantage, and UHC Policy Factors Every Provider Must Know in 2026
The policy landscape for wound graft coverage has shifted meaningfully in the last 18 months. Understanding what changed — and how those changes affect your authorization and billing workflow — is the difference between practices that maintain strong collection rates and those that are absorbing unexpected write-offs.
CMS 2026 Final Rule — Skin Substitute Reclassification
The most consequential structural change in recent years is CMS’s reclassification of skin substitute products under OPPS rulemaking into high-cost and low-cost payment categories. This replaced the previous pass-through payment status and bundled packaging approach that many products operated under.
What this means in practice for outpatient wound care billing:
- High-cost skin substitutes now carry a separate OPPS payment — CPT status indicator T
- Add-on codes (15272, 15274, 15276, 15278) carry status indicator N — they are packaged into the primary procedure APC and generate no separate payment
- ASC billing note: Add-on codes at ASCs carry payment indicator N1 — do not bill these separately expecting reimbursement
APC assignments from the AmnioRepair Coding Guide (reflecting current OPPS structure):
| CPT Code | Procedure Description | APC | ASC Payment Indicator |
|---|---|---|---|
| 15271 | Skin sub graft, trunk/arms/legs, first 25 sq cm | APC 5054 (Level 4 Skin) | G2 |
| 15273 | Skin sub graft, trunk/arms/legs, ≥100 sq cm, first 100 sq cm | APC 5055 (Level 5 Skin) | G2 |
| 15275 | Skin sub graft, face/scalp/hands/feet, first 25 sq cm | APC 5054 | G2 |
| 15277 | Skin sub graft, face/scalp/hands/feet, ≥100 sq cm, first 100 sq cm | APC 5054 | G2 |
| 15272, 15274, 15276, 15278 | Add-on codes (each additional sq cm group) | Status N (packaged) | N1 |
The reclassification also affects how payers set their coverage criteria. When CMS restructures reimbursement, payers use it as an opportunity to tighten clinical criteria for higher-cost products. The practical result is that products in the high-cost category face stricter PA scrutiny in 2026 than they did under the previous pass-through payment structure.
UHC Medicare Advantage — The Three Facts Every Wound Care Biller Must Know
From UHC’s clinical FAQ (Policy PCA-1-24-02596):
Fact 1: CMS has no National Coverage Determination (NCD) for non-porcine skin substitutes. This means LCDs govern where they exist — and in states without an active LCD, UHC applies their own Commercial and Individual Exchange Medical Policy for Skin and Soft Tissue Substitutes. Knowing which policy applies in your state is a prerequisite for building the right documentation package.
Fact 2: UHC’s review is post-service, pre-pay — not pre-service. The authorization step for these plans happens after claim submission, not before the procedure. This does not eliminate documentation requirements; it relocates them. Your documentation must be complete before you apply the graft, so it’s ready to send when UHC requests it after billing.
Fact 3: Determinations go back to providers through the UHC Provider Portal, not by fax or mail. If you’re not actively monitoring the portal, you’ll miss records requests and hit the response deadline without realizing it. Assign someone in your billing team to check the portal daily for any skin substitute claims under review.
LCD Coverage by MAC Jurisdiction — 2026 Reference
| MAC | Jurisdiction | Active LCD | States/Territories |
|---|---|---|---|
| CGS Administrators | J15 | L36690 — CTPs Lower Extremities | KY, OH |
| Novitas Solutions | JH / JL | L35041 — Bioengineered Skin Substitutes | AR, CO, DC, DE, LA, MD, MS, NJ, NM, OK, PA, TX |
| First Coast Service Options | JN | L36377 — Skin Substitutes DFU/VLU | FL, PR, VI |
| WPS / Palmetto / NGS | Various | LCD varies per MAC | IA, KS, MO, NE, IN, MI, NC, SC, VA, WV, IL, MN, WI, CT, ME, MA, NH, NY, RI, VT |
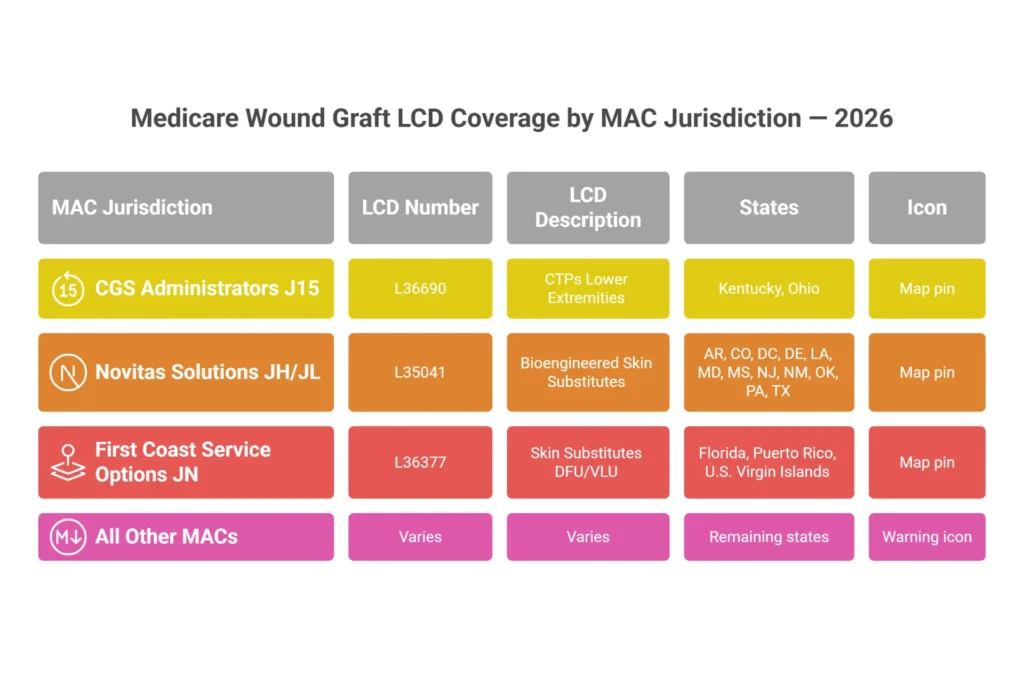
2026 provider note: In states without an active LCD, your MAC applies broader medical necessity criteria that aren’t codified in a coverage document. This increases audit exposure because there’s no published checklist to follow. In these states, build documentation to the most rigorous LCD standard available nationally — typically L35041 or L36377 — rather than assuming less documentation is acceptable. Our Medicare wound care billing guide has the complete state-by-state LCD mapping.
MS-DRG Coding for Inpatient Wound Graft (Hospital Billing Teams)
For hospital inpatient wound graft billing, the revenue recognition model works differently. MS-DRG assignment governs total reimbursement — not individual CTP codes. Understanding which DRGs apply to your skin graft encounters is essential for hospital billing staff.
Relevant MS-DRGs from the AmnioRepair Coding Guide:
- MS-DRG 463/464/465: Wound Debridement and Skin Graft — Musculo-Connective Tissue Disorders (with/without MCC or CC)
- MS-DRG 573/574/575: Skin Graft for Skin Ulcer or Cellulitis (with/without MCC or CC)
- MS-DRG 622/623/624: Skin Grafts and Wound Debridement — Endocrine/Nutritional/Metabolic Disorders
- MS-DRG 904/905: Skin Grafts for Injuries
ICD-10-PCS hospital inpatient procedure coding for skin graft replacement procedures uses: Section Ø (Medical and Surgical), Body System H (Skin and Breast), Root Operation R (Replacement), Device K (Nonautologous Tissue Substitute), Qualifier 4 (Partial Thickness).
For providers managing both outpatient wound care and hospital-based wound programs, the coding framework differs substantially between the two settings. Our medical coding services team manages both environments.
How GRAFT TRACK Eliminates Prior Authorization Delays for Wound Care Practices
Prior authorization for wound care grafts is genuinely one of the most time-intensive administrative tasks in a wound care practice. The documentation gathering, form completion, payer portal navigation, status tracking, records request response, and appeal management — when you add it up across all the graft applications in a week, it’s a meaningful portion of a billing or front-office staff member’s time.
GRAFT TRACK is EliteMedFinancials’ Verified Graft Program, built specifically around this problem. Here’s exactly what it covers and how it fits into your workflow.
What GRAFT TRACK Handles in the Prior Authorization Step
Prior authorization is Step 3 in the GRAFT TRACK workflow — after patient eligibility verification and benefits verification, and before clean claim submission. Every step has a defined owner and a defined checklist:
Step 1: Patient eligibility verification — Confirm active coverage, plan type, and deductible/coinsurance status before the first appointment.
Step 2: Benefits verification + product formulary check — Confirm the specific product is covered, determine whether PA is pre-service or post-service, calculate out-of-pocket liability.
Step 3: Prior authorization (wound graft specific) — This is where GRAFT TRACK’s core PA service operates:
- Complete payer-specific PA forms for all payer types including MA, commercial, D-SNP, and Tricare
- Documentation pre-review — your file is checked against a 14-point documentation checklist before submission to catch gaps before the payer does
- Portal submission with timestamp and tracking
- Daily status monitoring — follow-up on every open authorization request
- Records request response management — immediate notification and 5-business-day turnaround guaranteed
- Authorization number entered into your billing system before claim submission
- Appeal management for any denied PA — first and second level, with physician LMN support
Step 4: Clean claim submission — CPT, Q-code, modifiers, Field 23, Field 19 — all completed by specialists who handle wound graft billing daily.
Step 5: Denial management and AR — Any post-submission denials go to the denial management team for analysis, correction, and resubmission.
What Practices Experience When Prior Auth Is Handled Correctly
The difference between a practice with a structured PA workflow and one without it shows up in these numbers:
- Zero retroactive denials for missing prior authorization — because the auth step happens before the clinical visit, not after
- No write-offs from missed product formulary checks — because the formulary confirmation happens at verification, not at billing
- No claim holds from UHC MA post-pay review — because documentation is pre-verified and ready to submit within 48 hours of any records request
- Reduced billing staff time on PA rework — average 6–8 hours per week recovered for practices doing 10+ graft applications per week
- Predictable approval timelines communicated to clinical scheduling — clinical staff knows whether to book the graft appointment before or after authorization clears
This is the operational outcome that wound care practices with high graft volumes have been building toward. GRAFT TRACK delivers it as a managed service — which means your clinical staff focuses on wound care, and your administrative team focuses on patient experience, not insurance paperwork.
💼 Ready to Stop Writing Off Missed Prior Authorizations?
Wound care graft prior authorization is handled end-to-end in GRAFT TRACK’s Verified Graft Program — from pre-application insurance verification through authorization, clean claim submission, and denial management. No more chasing payer portals. No more missed records deadlines. No more retroactive write-offs.
🏥 Already Seeing Denials on Wound Graft Claims?
Our team reviews your denied claims, identifies documentation gaps, rebuilds the appeal package, and manages all levels of the appeals process. Most wound graft denials are recoverable with the right approach and the right documentation.
Quick Reference Table — Wound Care Graft Prior Authorization 2026
| Topic | Key Reference |
|---|---|
| Primary CPT codes | 15271, 15272, 15273, 15274, 15275, 15276, 15277, 15278 |
| Common Q-codes | Q4101 Apligraf · Q4106 Dermagraft · Q4131 EpiFix · Q4115 AmnioFix · Q4235 AmnioRepair · Q4204 Kerecis |
| Required modifiers | JC (graft used) · JW (wastage) · JZ (no waste) · 59/XS (distinct procedure) |
| Ocular amniotic CPT | 65778 (placement without sutures — separate pathway from wound codes) |
| Key ICD-10 codes | E11.621 · E10.621 · I83.009 · L89.xxx (pressure) · L97.xxx (non-pressure) |
| Governing LCDs | L36690 (CGS / KY-OH) · L35041 (Novitas) · L36377 (First Coast) |
| CMS-1500 PA field | Field 23 — must contain auth number before claim submission |
| CMS-1500 product field | Field 19 — product name, NDC/UPC/HRIC, invoice price |
| UHC MA review type | Post-service, pre-pay clinical review (non-porcine products) |
| UHC Provider contact | 888-676-7768 (UHC) · 800-873-4575 (Optum) |
| ProCare Advantage fax | 1-833-610-2399 |
| ProCare portal | secure.healthx.com/ProCareAdvantage.Provider |
| Standard PA turnaround | 3–5 business days (routine) · 24–72 hours (urgent) |
Voice Search: Wound Care Graft Prior Authorization Questions Answered
Common questions asked conversationally — direct answers optimized for featured snippets.
Does Medicare require prior authorization for wound grafts?
Original Medicare Part B does not require pre-service prior authorization for wound graft procedures. However, Medicare Administrative Contractors conduct post-payment audits on claims, and any claim that doesn’t meet the applicable LCD criteria can be denied retroactively — even years after the service date. Medicare Advantage plans are a different matter: virtually all MA plans require prior authorization for skin substitute applications, with requirements varying by plan type and carrier.
How long does wound graft prior authorization take?
Standard pre-service prior authorization for wound grafts takes 3 to 5 business days for routine requests. Urgent requests classified as serious jeopardy to the patient’s health are processed in 24 to 72 hours. For UHC Medicare Advantage plans using a post-service review model, the review occurs after claim submission — not before the procedure — and can take 10 to 30 days for a determination.
What CPT codes require prior authorization for wound grafts?
CPT codes 15271 through 15278 are the primary skin substitute application codes that trigger prior authorization review. Each of these codes must be paired with the product’s Q-code or HCPCS code — for example, Q4235 for AmnioRepair or Q4131 for EpiFix. The Q-code identifies the specific product to the payer, who then verifies it against their covered product formulary before approving or denying the authorization.
What is the most common reason wound graft prior authorization is denied?
The most common reason is insufficient conservative care documentation — meaning the medical record doesn’t show at least four consecutive weeks of standard wound treatment before the graft was requested. Other frequent denial reasons include missing vascular assessments for diabetic foot ulcers, the selected product being excluded from the plan’s covered formulary, and incomplete wound measurements. Almost all of these denials are preventable with a structured pre-submission documentation review.
Is CPT 65778 for ocular amniotic membrane covered by insurance?
CPT 65778, which covers placement of an amniotic membrane on the ocular surface without sutures, is covered under a separate authorization pathway from wound application codes. Covered indications typically include neurotrophic keratitis, corneal ulcers, Stevens-Johnson syndrome, persistent epithelial defects, and severe dry eye (DEWS grade 3–4) that have not responded to earlier stages of treatment. This code has its own ophthalmic LCD criteria and requires separate documentation that should never be confused with wound graft PA templates.
Can prior authorization for a wound graft be appealed after a denial?
A denied wound graft prior authorization can absolutely be appealed. There are typically three levels available: a first-level internal reconsideration within 30 days, a second-level independent review if the first appeal fails, and for Medicare Advantage patients, the full five-level Medicare appeals process — which includes an Administrative Law Judge hearing for claims over $180 and extends to Federal District Court. The key to a successful appeal is addressing the specific denial reason with targeted additional documentation, not resubmitting the same package that was already denied.
Which insurance plans have the highest approval rates for amniotic graft prior auth?
Approval rates depend more on documentation quality than on the specific carrier. Plans that use post-service review rather than pre-service PA — like some UHC Medicare Advantage products — tend to have higher initial pass-through rates, but claims can still be clawed back if documentation doesn’t hold up under review. Porcine-based products consistently face lower PA friction across commercial and MA plans compared to non-porcine amniotic and cellular products. A clean, complete documentation package aligned with LCD criteria gives you the best approval odds regardless of carrier.
FAQ: Detailed Answers for Billing Teams and Wound Care Providers
Which wound graft products most commonly require prior authorization?
Cellular tissue-based products (CTPs) and amniotic membrane products trigger prior authorization most consistently across payer types. Products like Apligraf (Q4101), EpiFix (Q4131), AmnioRepair (Q4235), Dermagraft (Q4106), and Grafix require pre-service or post-service authorization from virtually all Medicare Advantage, commercial, and D-SNP plans. Porcine-derived acellular products like Oasis Wound Matrix face lower authorization scrutiny under UHC’s policy, which specifically applies enhanced clinical review to non-porcine skin substitutes. For any new product entering your wound care formulary, confirm its PA requirement during initial product credentialing before the first patient application.
Can you bill CPT 15271 and 15275 together on the same claim?
CPT 15271 (skin substitute graft, trunk/arms/legs, first 25 sq cm) and CPT 15275 (skin substitute graft, face/scalp/hands/feet, first 25 sq cm) can appear on the same claim only when a patient has distinct wounds in both anatomical groups. These codes represent separate anatomical regions — wounds within each group are totaled together, while wounds in different groups are billed on separate lines with different CPT codes. If a patient has a diabetic foot ulcer (CPT 15275 territory) and a leg wound (CPT 15271 territory), billing both codes together is appropriate and accurate. Billing both for wounds in the same anatomical region would be a coding error.
Does CPT 15271 include debridement?
Yes. CPT coding guidance specifies that skin substitute graft application codes 15271 through 15278 include simple tissue debridement performed as part of wound bed preparation. Simple debridement that is integral to applying the graft is not separately reportable. If extensive or complex debridement was performed as a distinct, separately documented service — not simply wound bed preparation for the graft — it may be separately reportable with careful documentation of the clinical distinction. When in doubt, document the debridement separately in the record even if you don’t bill it separately — reviewers look for that documentation.
What is the JW modifier and when is it used for wound grafts?
The JW modifier identifies drug or biological wastage — the portion of a product that was opened but not used on the patient. For wound grafts, this applies when a graft product is opened but only partially used. For example, if a 4×6 cm AmnioRepair sheet is opened but only 4×4 cm (16 sq cm) is applied to the wound, the remaining 8 sq cm is wasted. Bill the used portion with Q4235 + JC modifier; bill the wasted portion on a separate line with Q4235 + JW modifier. When the entire opened product is applied with no waste, use the JZ modifier to document zero wastage. CMS and MA plans audit JW/JZ usage — discrepancies between lot documentation and billing records trigger payment recovery reviews.
What documentation does UHC Medicare Advantage require for skin substitute claims?
UHC Medicare Advantage applies post-service, pre-pay clinical review for non-porcine skin substitute claims under Policy PCA-1-24-02596. When documentation is requested after claim submission, providers must supply medical records supporting all medical necessity criteria from the applicable LCD — including wound type, wound duration, conservative treatment history with visit dates and treatments used, wound measurements from initial and current visits, vascular assessment results, and evidence that the specific product selected meets the coverage criteria for that wound type. Providers are notified through the UHC Provider Portal; members receive notice by mail. Failure to respond results in denial. Appeal instructions are included in all denial notices. Provider contact for appeals: 888-676-7768 (UHC) or 800-873-4575 (Optum).
What ICD-10 codes support wound graft prior authorization?
The three most commonly used ICD-10 codes in wound graft PA requests are E11.621 (Type 2 diabetes with foot ulcer), I83.009 (varicose veins with ulcer, lower extremity), and the L89.xxx series for pressure injuries (where the final characters specify anatomical site and stage). Additional codes used include E10.621 (Type 1 DM foot ulcer), E11.622 (Type 2 DM other skin ulcer), and L97.xxx codes for non-pressure chronic ulcers of the lower extremity. The ICD-10 code on your PA request must map to a covered wound type under the applicable LCD — submitting a code that’s not on the LCD’s covered indications list is a near-certain denial. For a complete wound care ICD-10 and CPT code reference, see our wound care billing codes guide.
How does amniotic graft prior auth differ from standard skin substitute PA?
Amniotic membrane graft prior authorization follows the same foundational framework as other CTPs — LCD compliance, wound type criteria, conservative care documentation — but carries additional product-specific scrutiny because amniotic products tend to be among the highest-cost items in the wound care billing category. Payers often require documentation of why an amniotic product was selected over a lower-cost alternative. You’ll need to include the lot number, the invoice price, and in some cases peer-reviewed clinical evidence supporting amniotic membrane use for the specific wound presentation. For ocular amniotic membrane applications under CPT 65778, the pathway is entirely separate — using ophthalmic conservative treatment step sequences and ocular-specific indications that have no overlap with wound application documentation. Never cross-use documentation templates between wound and ocular authorization requests.
Does outsourcing wound care billing improve prior authorization outcomes?
It typically does — for a specific reason. Specialized wound care billing teams process authorization requests for the same payer types and product categories repeatedly, which means they recognize the documentation gaps that trigger denials before submission, not after. They also maintain current knowledge of payer policy changes, formulary updates, and LCD revisions that affect authorization requirements. In-house billing staff managing authorization alongside scheduling, coding, posting, and denial management often miss the incremental policy changes that accumulate into recurring denial patterns. For practices doing significant graft volume, the ROI from reduced write-offs and recovered denial revenue typically covers the cost of outsourced billing services many times over. Read our full outsource wound care billing guide for a detailed framework.
Conclusion: Getting Wound Care Graft Prior Authorization Right in 2026
After reading this guide, you understand something that most wound care providers only figure out through expensive experience: prior authorization for wound grafts is not an administrative nuisance. It is a clinical and financial workflow that — when done right — protects your revenue, protects your patients from unexpected liability, and keeps your practice out of post-pay audit territory.
The requirements are demanding. The documentation checklist is long. Payer policies vary in ways that create real operational complexity — UHC Medicare Advantage doesn’t work like traditional Medicare, ProCare Advantage doesn’t work like Aetna commercial, and the product-specific formulary requirements layer on top of all of it. But every step in this process is learnable, systematizable, and manageable — with the right workflow and the right team behind it.
If your practice is handling wound graft prior authorization in-house with general billing staff and no wound-graft-specific protocol, the gap between where you are and where your collection rates could be is often measured in tens of thousands of dollars per year. That’s not a criticism — it’s an opportunity.
🔵 Ready to Eliminate Prior Auth Delays and Protect Your Graft Revenue?
GRAFT TRACK’s Verified Graft Program handles every step — insurance verification, product formulary check, prior authorization, clean claim submission, and denial management — as a complete managed service built specifically for wound care practices.
No more retroactive write-offs. No more missed records deadlines. No more chasing payer portals.
🟢 Already Getting Denied on Wound Graft Claims?
Retroactive denials and post-pay recoupment are recoverable — when the right team handles the appeal. Our denial management specialists review denied wound graft claims, identify the specific gaps, and build appeal packages that address the exact criteria the payer cited.
🔴 New to Wound Graft Billing?
Start with our complete coverage and billing library before your first graft claim:
EliteMedFinancials.com provides wound care billing, coding, RCM, and credentialing services for wound care practices, podiatry groups, and hospital-based wound care programs. Our complete RCM services and end-to-end financial management, including enrollment, digital marketing, and IT services, are purpose-built for healthcare providers who want their administrative infrastructure to work as hard as their clinical team.
External Resources:
- CMS LCD Database — MAC Coverage Policies — Search for current LCD policies by state and product category
- AMA CPT Code Set Guidelines — Official CPT coding guidelines for skin substitute procedures

