Here’s a number that should stop you cold: the denial rate for amniotic membrane graft claims runs between 25 and 35 percent — nearly three times the national average for other procedures. For a wound care practice or ophthalmology group doing moderate volume, that’s not a billing problem. That’s a revenue crisis happening in slow motion.
The frustrating part? Most of those denials are preventable. They come down to wrong CPT code selection, mismatched Q-codes, missing documentation, and medical necessity criteria that nobody explained clearly when your practice started offering these procedures.
This guide fixes that. You’ll get a complete, updated reference for amniotic membrane graft billing — every CPT code, every relevant Q-code with actual Medicaid fee data, Medicare’s LCD requirements, ICD-10 combinations that work, and a step-by-step workflow your billing team can follow today. Whether you’re managing wound care applications for diabetic foot ulcers or billing for ocular surface procedures in an ophthalmology practice, this is the only resource you’ll need.
Here’s what you’ll walk away with:
- Exact CPT codes for ocular (65778–65780) and wound care (15271–15278) amniotic graft procedures
- Verified Medicaid Q-code reimbursement data for 14+ products
- A step-by-step billing workflow that catches denials before submission
- Medicare LCD requirements and commercial payer differences
- Documentation checklists built for RAC audits
- ICD-10 codes — including aftercare codes and ICD-10-PCS for inpatient
- A denial appeal strategy organized by denial type
Table of Contents
What Is Amniotic Membrane Graft Billing?
Amniotic membrane graft billing is the process of coding, documenting, and submitting claims for procedures involving human placental tissue — specifically the innermost layer of the placenta — applied to promote wound healing or reconstruct damaged ocular surfaces. It’s a two-code system: one CPT code for what you did, one HCPCS Q-code for what you used. Get either one wrong, and your claim is going nowhere.
Clinically, these grafts earn their price tag. Amniotic tissue contains a cocktail of growth factors (EGF, TGF-α, TGF-β, PDGF), anti-inflammatory cytokines, and an extracellular matrix scaffold that does three things conventional wound care can’t: it suppresses inflammation at the wound site, inhibits scar formation, and provides a structural template that guides the patient’s own cells to regenerate tissue. The clinical outcomes are compelling — randomized controlled trials show EpiFix achieving 62% healing in 12 weeks for diabetic foot ulcers versus 8% in standard care controls.
The billing challenge is that these products are expensive, heavily audited, and governed by Local Coverage Determinations (LCDs) that require proof of failed standard care before you apply a single square centimeter. Add in product-specific Q-codes, wound-size-based code selection, Medicare’s 2026 flat-rate transition, and commercial payer variations — and you have one of the most complex billing workflows in modern outpatient medicine.
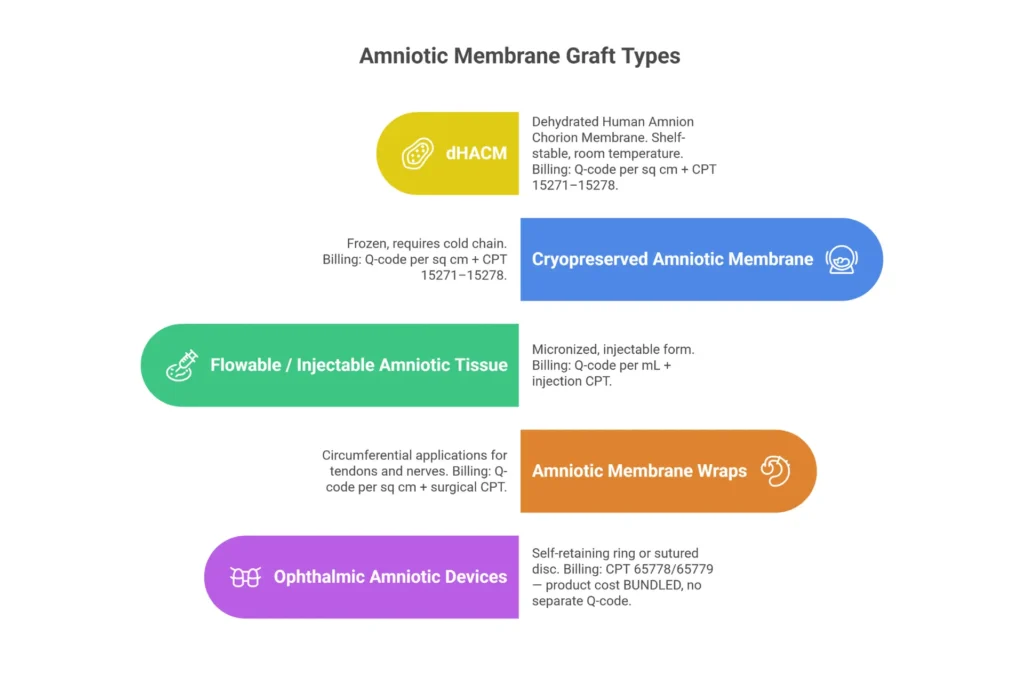
Types of Amniotic Membrane Grafts Used in Clinical Practice
Not all amniotic products bill the same way. The processing method determines the Q-code, and the Q-code determines your reimbursement.
| Product Type | Description | Billing Classification | Representative Products |
|---|---|---|---|
| dHACM (Dehydrated) | Shelf-stable, room temperature storage, extended shelf life | Per sq cm Q-codes | EpiFix (Q4186), AmnioBand (Q4191), BioDfence (Q4188) |
| Cryopreserved | Frozen, retains higher bioactivity, requires cold chain | Per sq cm Q-codes | Grafix Core (Q4133), Biovance (Q4184) |
| Flowable/Injectable | Micronized amniotic tissue for injection | Per mL/mg Q-codes | VIAFLO (Q4263), AmnioFix Injectable |
| Wraps | Circumferential application for tendons and nerves | Per sq cm Q-codes | AmnioWrap2 (Q4271), AlloWrap (Q4205) |
| Ophthalmic Devices | Self-retaining rings or sutured grafts for ocular surface | Bundled into CPT | Prokera (CPT 65778), AmnioGraft (CPT 65779) |
One thing practices often miss: the billing treatment of ophthalmic devices is fundamentally different from wound care products. For Prokera and similar devices, the product cost is bundled into the CPT payment — you do not bill a separate Q-code. For wound care products, the Q-code is billed separately, per square centimeter, in addition to the application CPT code.
Which Specialties Bill for Amniotic Membrane Graft Procedures?
Amniotic graft billing spans more specialties than most coders realize:
- Wound Care Specialists — chronic venous leg ulcers, diabetic foot ulcers, pressure injuries, post-surgical wounds
- Ophthalmologists — dry eye disease (DEWS Stage 3/4), neurotrophic keratitis, corneal ulcers, pterygium, chemical burns, Stevens-Johnson syndrome
- Podiatrists — diabetic foot ulcers, heel wounds, post-surgical wound care
- Orthopedic Surgeons — tendon wraps, nerve protection at surgical sites, soft tissue reconstruction
- Plastic Surgeons — burn reconstruction, complex wound coverage, scar revision
- Dermatologists — non-healing surgical wounds, biopsy sites, skin cancer reconstruction
Each specialty has its own coding nuances, documentation requirements, and payer patterns. A podiatrist billing for a DFU follows a different pathway than an ophthalmologist billing for neurotrophic keratitis — and confusing those pathways is a reliable way to generate denials. We’ll walk through each in Section 4.
CPT Codes for Amniotic Membrane Graft — Complete 2025–2026 Reference
The CPT code you select for an amniotic graft claim does two jobs: it tells the payer what procedure you performed and which reimbursement rate applies. There are two separate CPT families — ophthalmic codes (65778–65780) and skin substitute application codes (15271–15278) — and they are not interchangeable.
Master CPT Code Reference Table — Amniotic Membrane Graft Billing 2025–2026
Table 1: Complete CPT Code Reference for Amniotic Membrane Graft Billing — Wound Care and Ophthalmology
| CPT Code | Official Description | Specialty | Global Period | 2025 Medicare NF Rate* | Key Modifiers | Primary ICD-10 |
|---|---|---|---|---|---|---|
| 65778 | Placement of amniotic membrane on ocular surface; without sutures | Ophthalmology | 10-day | ~$465 | RT/LT, -50, -59 | H16.233, H16.009 |
| 65779 | Placement of amniotic membrane on ocular surface; with sutures | Ophthalmology | 10-day | ~$500 | RT/LT, -50, -59 | H11.001, H16.041 |
| 65780 | Ocular surface reconstruction; amniotic membrane transplantation | Ophthalmology | 90-day | ~$1,200 | RT/LT, -59 | T26.60XA, H18.52 |
| 15271 | Skin substitute graft, trunk/arms/legs; first 25 sq cm or less | Wound Care | 0-day | ~$250 | -59, -51, RT/LT | E11.621, L97.419 |
| 15272 | Skin substitute graft, trunk/arms/legs; each add’l 25 sq cm (add-on) | Wound Care | N/A | Per unit | -59 | Same as 15271 |
| 15273 | Skin substitute graft, trunk/arms/legs; first 100 sq cm or less | Wound Care | 0-day | ~$350 | -59, -51, RT/LT | L89.039, I83.011 |
| 15274 | Skin substitute graft, trunk/arms/legs; each add’l 100 sq cm (add-on) | Wound Care | N/A | Per unit | -59 | Same as 15273 |
| 15275 | Skin substitute graft, face/scalp/eyelids/neck; first 100 sq cm | Wound Care | 0-day | ~$350 | RT/LT, -59, -51 | T20.00XA, L89.219 |
| 15276 | Skin substitute graft, face/scalp/eyelids; each add’l 100 sq cm (add-on) | Wound Care | N/A | Per unit | RT/LT, -59 | Same as 15275 |
| 15277 | Skin substitute graft, hands/feet/genitalia; first 100 sq cm | Wound Care / Podiatry | 0-day | ~$350 | RT/LT, -59, -51, Q8 | E11.621, L97.419 |
| 15278 | Skin substitute graft, hands/feet; each add’l 100 sq cm (add-on) | Wound Care / Podiatry | N/A | Per unit | RT/LT, -59 | Same as 15277 |
Non-facility rates based on 2025 Medicare Physician Fee Schedule; actual rates vary by geographic locality. Verify current rates at CMS.gov PFS Look-Up Tool.

CPT 65778 — What It Covers and Medical Necessity Requirements
CPT 65778 captures self-retaining amniotic membrane placements — ring-based devices like Prokera that sit on the ocular surface and release therapeutic proteins over 5–14 days as the membrane gradually dissolves. No sutures, no operating room, but don’t underestimate the documentation requirements.
Medicare medical necessity checklist for 65778:
- Diagnosis of persistent epithelial defect, corneal ulcer, neurotrophic keratitis, severe dry eye (DEWS Stage 3/4), or recurrent corneal erosion
- Documented failure of at least 4 weeks of conservative therapy: preservative-free artificial tears (minimum QID), ointments, punctal plugs, Restasis or Xiidra
- Measured corneal staining grade ≥3 using standard grading scale
- Defect dimensions documented in chart (length × width in mm)
- No active infection, neoplasm, or contraindication to amniotic tissue
- Color photographs with slit lamp scale visible, pre- and post-placement
The 10-day global period is your other watchout: all post-operative visits during days 1–10 are included in the procedure payment. Bill a separate E/M only when a new, unrelated problem arises during that window — and document clearly why the visit was distinct.
CPT 65779 vs. CPT 65778 — Key Differences
These two codes trip up even experienced coders because the clinical distinction sounds minor but has real billing consequences.
| CPT 65778 | CPT 65779 | |
|---|---|---|
| Fixation Method | Self-retaining ring device (Prokera, BioTissue) | Sutured or fibrin-glued directly to ocular surface |
| Typical Indications | Dry eye, neurotrophic keratitis, persistent epithelial defects, mild-moderate burns | Larger defects, unstable surface requiring fixation, severe burns |
| Setting | Office or ASC | Typically ASC or hospital outpatient |
| 2025 Medicare NF Rate | ~$465 | ~$500 |
| Global Period | 10 days | 10 days |
| Documentation Key | “Self-retaining amniotic membrane placed without sutures” | “Amniotic membrane graft sutured with 10-0 nylon at [clock positions]” |
What is the difference between 65778 and 65779? The fixation method. 65778 is self-retaining (no sutures). 65779 requires sutures or glue for fixation. The indication — not the product name — should drive code selection.
Does CPT 65778 have a global period? Yes, 10 days.
CPT 65780 — Ocular Surface Reconstruction
CPT 65780 is in a different category entirely. This code describes true ocular surface reconstruction — typically multiple layers of amniotic membrane used to rebuild a severely compromised corneal surface in conditions like limbal stem cell deficiency, extensive chemical burns, or Stevens-Johnson syndrome. The 90-day global period and ~$1,200 reimbursement reflect the complexity.
Don’t use 65780 for routine dry eye or corneal ulcer management. It’s reserved for cases where the goal is structural reconstruction of the ocular surface, often performed under general or monitored anesthesia in a surgical setting.
CPT Codes 15271–15278 — Skin Substitute Graft Application
For wound care, your coding lives in this family. Code selection depends on two variables working together:
Variable 1 — Anatomical location:
- Trunk, arms, legs → 15271 (first 25 cm²) or 15273 (first 100 cm²)
- Face, scalp, eyelids, neck → 15275 (first 100 cm²)
- Hands, feet, genitalia → 15277 (first 100 cm²)
Variable 2 — Total wound surface area:
- Trunk/arm/leg wounds ≤25 cm² → 15271 (no add-on needed)
- Trunk/arm/leg wounds 26–50 cm² → 15271 + 15272 ×1
- Trunk/arm/leg wounds >100 cm² → 15273 (base) + 15274 (add-ons per 100 cm²)
- Face/foot wounds ≤100 cm² → 15275 or 15277 alone
- Face/foot wounds >100 cm² → 15275 or 15277 + add-on per additional 100 cm²
How to Bill CPT 15275 (and Can You Bill 15004 and 15275 Together?)
How to bill CPT 15275:
- Debride the wound first if indicated (document separately — 97597 or 97598 for whirlpool/sharp selective debridement)
- Measure wound area post-debridement (length × width = cm²)
- Bill 15275 once for the first 100 sq cm of the face/scalp/eyelid/neck wound
- Add 15276 for each additional 100 sq cm
- Separately bill the Q-code × total cm² of graft applied
Can you bill 15004 and 15275 together? Generally, no. The NCCI bundles surgical preparation (15004) into the application code. The exception is when you perform surgical preparation on one wound and apply a graft to a completely separate wound at a distinct anatomical site on the same date of service — then modifier -59 applies, but document the distinct sites explicitly in your notes.
CPT Code for Amniotic Membrane Graft on the Foot
CPT 15277 is your code for foot wounds. Hands, feet, and genitalia are grouped together in this family regardless of whether the foot wound is on the dorsum, plantar surface, or heel. Use 15278 as the add-on for each additional 100 sq cm beyond the first 100.
⚠️ Common Billing Mistake: A common error is using 15271 (trunk/extremity code) for foot wounds. The foot falls under 15277, not 15271. This mismatch will trigger a denial every time, because payers cross-reference the CPT code against the anatomical site documented in the procedure note.
🔵 Need Help Selecting the Right CPT Codes for Your Amniotic Graft Claims?
Code selection errors are the #1 preventable cause of amniotic graft denials. Our certified wound care billing specialists review your coding, documentation, and payer contracts to make sure you’re getting paid for every procedure.
Get a Free Coding Review from Elite Med Financials →
HCPCS Q-Codes for Amniotic Graft & Wound Care Skin Substitutes
Here’s how to think about Q-codes: the CPT code tells your payer what you did. The Q-code tells your payer exactly which product you used. For amniotic membrane grafts, both codes are mandatory on most claims — and they have to match your product documentation or you’ll be explaining yourself in a RAC audit.
What Are Q-Codes? (A Definition for Medical Billers)
What are Q-codes in billing? Q-codes are temporary HCPCS Level II codes in the Q0000–Q9999 range. CMS assigns them to emerging biologics and supplies that don’t yet have permanent CPT or HCPCS codes. They’re product-specific — Q4186 identifies EpiFix by MiMedx, while Q4133 identifies Grafix Core by Smith+Nephew. The specificity matters enormously: acquisition costs for amniotic products range from around $400 per sq cm all the way to $4,600, and payers need to know exactly what you’re billing before they price the claim.
Unlike CPT codes maintained by the AMA, Q-codes are assigned and updated by CMS on a quarterly schedule. Products can get new codes, have codes revised, or transition from temporary Q-codes to permanent HCPCS codes as evidence matures.
Does Medicare cover Q-codes? Yes — when billed alongside appropriate CPT application codes and supported by documentation meeting LCD medical necessity criteria. In 2026, CMS moved most non-BLA (Biologics License Application) amniotic products to a flat rate of $127.28 per sq cm in office settings under the Physician Fee Schedule, which significantly compresses margins compared to pre-2026 ASP+6% payment.
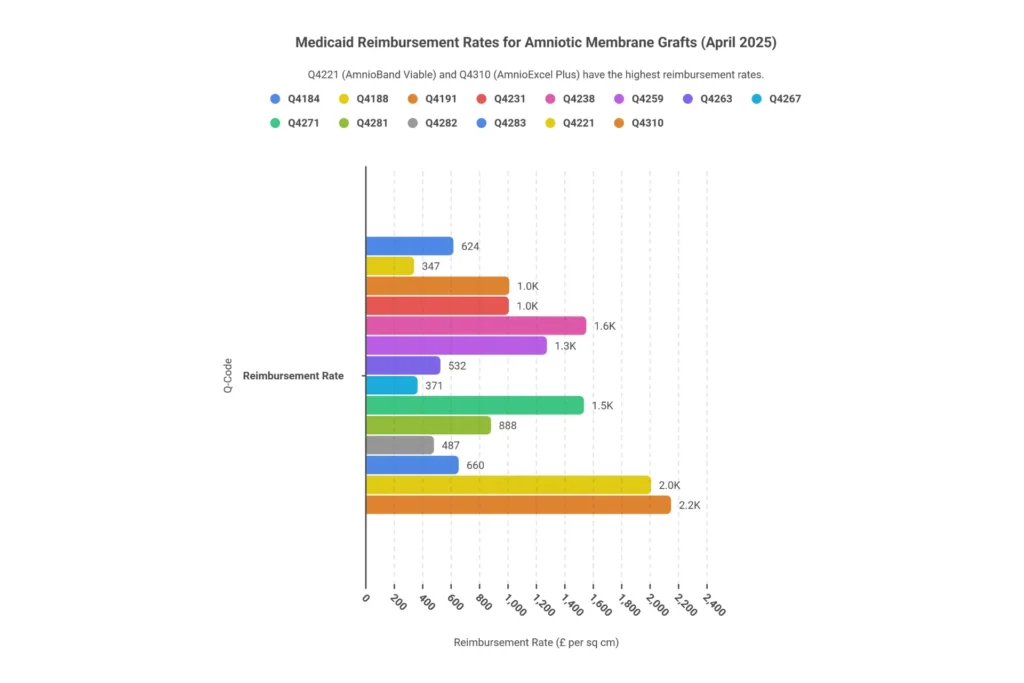
Complete Q-Code List and Medicaid Reimbursement Rates (Verified April 2025)
The table below reflects verified Medicaid fee data as of April 1, 2025. Medicaid rates typically run 50–70% of Medicare ASP rates and vary by state — use this as a baseline for comparison and appeals.
Table 2: Q-Code Reimbursement Reference — Amniotic Membrane Products, Wound Care Skin Substitutes 2025–2026
| Q-Code | Product Description | Brand Name(s) | Form | Unit | Medicaid Fee (Apr 2025) | 2025 Medicare (ASP+6%) | Notes |
|---|---|---|---|---|---|---|---|
| Q4133 | Cryopreserved amniotic membrane | Grafix Core | Sheet | Per cm² | State-variable | ~$1,200 | Strong RCT data for DFU; requires cold chain |
| Q4173 | Porcine/amniotic composite graft | PuraPly AM | Sheet | Per cm² | State-variable | ~$650 | Antimicrobial properties; broad wound application |
| Q4184 | Cryopreserved amniotic membrane | Biovance | Sheet | Per cm² | $624.34 | ~$1,450 | ≤20 cm² initial MAC requirement |
| Q4188 | Dehydrated amniotic membrane | Amnioexcel, BioDfence | Sheet | Per cm² | $346.64 | ~$900 | Ophthalmic and podiatric applications |
| Q4191 | Dehydrated amnion/chorion membrane | AmnioBand, Guardian | Sheet | Per cm² | $1,016.31 | ~$2,300 | Tier 1 evidence; frequently prior-auth approved |
| Q4205 | Membrane wrap, per sq cm | AlloWrap (Stratus) | Wrap | Per cm² | State-variable | ~$800 | Circumferential; tendon/nerve applications |
| Q4221 | Cryopreserved viable amnion matrix | AmnioBand Viable | Sheet | Per cm² | $2,014.00 | ~$4,500 | High-cost; strict LCD enforcement; <100 cm² |
| Q4231 | Amnion/chorion membrane | EpiFix (MiMedx) | Sheet | Per cm² | $1,013.11 | ~$2,200 | 28% market share; strong DFU evidence |
| Q4238 | Dehydrated amniotic tissue | Amnioforce (Sanara) | Sheet | Per cm² | $1,557.99 | ~$3,400 | Viable cells; advanced wound protocol |
| Q4259 | Cryopreserved placental membrane | TissueXM | Sheet | Per cm² | $1,280.74 | ~$2,800 | Pressure ulcer applications |
| Q4263 | Injectable amniotic fluid allograft | VIAFLO | Flowable | Per mL | $531.78 | ~$1,100/mg | Joint and tendon injections; high denial risk |
| Q4267 | Placental membrane allograft | Reynard | Sheet | Per cm² | $371.31 | ~$850 | Lower cost tier; ortho and wound care |
| Q4271 | Layered amnion/chorion wrap | AmnioWrap2 | Wrap | Per cm² | $1,541.61 | ~$3,300 | Tendon wraps, surgical site reinforcement |
| Q4281 | Cryopreserved trilayer amniotic | Biovance Trilayer | Sheet | Per cm² | $888.11 | ~$1,900 | Advanced wound applications |
| Q4282 | Dehydrated human amnion/chorion | DHACM generic | Sheet | Per cm² | $486.78 | ~$1,050 | Tier 2 evidence; standard wound care |
| Q4283 | Amnion/chorion membrane wrap | XWrap | Wrap | Per cm² | $660.38 | ~$1,400 | Circumferential; ≤12 applications/year |
| Q4310 | Multi-layer amniotic membrane | AmnioExcel Plus | Sheet | Per cm² | $2,154.98 | ~$4,600 | Highest-cost tier; strict LCD criteria apply |
Source: CMS HCPCS data, verified April 2025. Medicare rates reflect ASP+6% for office settings; 2026 flat-rate transition at $127.28/cm² applies to most non-BLA products under MPFS.
Membrane Graft vs. Membrane Wrap — Billing Difference (Q4205)
Is Q4205 a membrane graft or membrane wrap per sq cm? Q4205 (AlloWrap by Stratus Medical) is a membrane wrap billed per square centimeter, designed for circumferential applications around tendons, nerves, or surgical sites. This is a fundamentally different product type than flat sheet grafts like Q4231 (EpiFix) that are applied directly to open wound beds.
Here’s why the distinction matters for billing:
- Flat sheet grafts (Q4184, Q4191, Q4231, etc.) — applied to open wounds; billed with CPT 15271–15278; requires wound measurement documentation
- Wraps (Q4205, Q4271, Q4283) — circumferential applications; may be billed with tendon repair or nerve repair CPT codes; often considered “integral to the primary procedure” by payers
Many payers scrutinize wrap applications in non-wound orthopedic contexts as investigational. If you’re billing wraps for tendon or nerve applications, prepare thorough appeals documentation citing peer-reviewed clinical evidence specific to the product.
💡 Expert Insight: The 2026 CMS flat-rate transition to $127.28/cm² applies primarily to non-BLA products under the Physician Fee Schedule in office settings. Hospital Outpatient settings (OPPS) may still reimburse via APC methodology. If your margins are being compressed under MPFS, evaluate whether shifting appropriate cases to hospital outpatient settings makes financial sense for your practice. Our RCM Services team can help you model that transition.
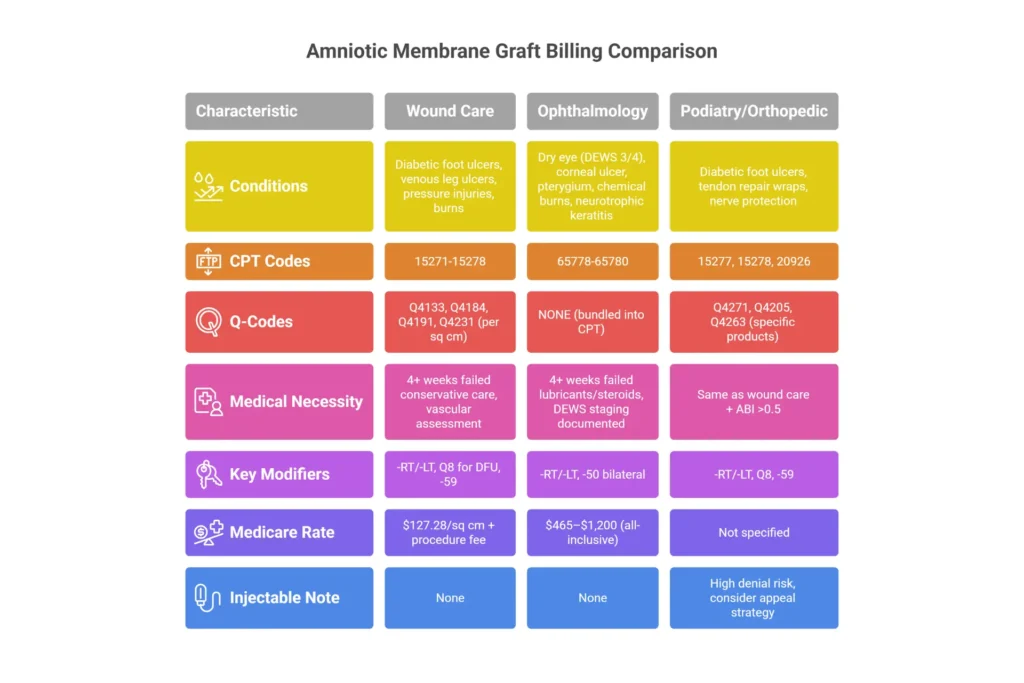
Amniotic Membrane Graft Billing: Wound Care vs. Ophthalmology
One of the fastest ways to generate denials is applying wound care billing logic to ophthalmic procedures — or vice versa. These are two different revenue ecosystems with different code families, different payment models, and different documentation standards.
| Parameter | Wound Care | Ophthalmology | Podiatry / Orthopedic |
|---|---|---|---|
| Primary Conditions | DFU, VLU, pressure ulcers, burns, post-surgical | Dry eye, corneal ulcer, pterygium, chemical burns, neurotrophic keratitis | DFU (foot), tendon repair, nerve wraps, surgical sites |
| CPT Codes | 15271–15278 | 65778–65780, 65426 | 15277–15278, 20926, 20999 |
| Q-Codes Billed | Q4133, Q4184, Q4191, Q4231, Q4238 | None billed separately (bundled) | Q4271, Q4205, Q4263 (high denial risk) |
| Payment Model | Product reimbursed separately per cm² | Product cost bundled into CPT rate | Per cm² for foot wounds; variable for wraps |
| 2026 Medicare Rate | $127.28/cm² (MPFS) + procedure fee | ~$465–$1,200 global (procedure only) | $127.28/cm² foot wounds |
| Key Modifiers | -59, -51, RT/LT, Q7, Q8, Q9 | RT/LT, -50, -59 | RT/LT, -59, Q7, Q8 |
| Medical Necessity Standard | 4+ weeks failed conservative care, vascular assessment | Failed lubricants/steroids, DEWS 3/4 staging | Same as wound care + ABI >0.5 for DFU |
| Prior Auth Required | ~80% commercial payers | Often waived for acute injuries | Required for most commercial |
| Typical Place of Service | 11 (office), 22 (HOPD), 31 (SNF) | 11 (office), 24 (ASC), 22 (HOPD) | 11 (office), 22 (HOPD) |
Wound Care Billing — Chronic Ulcers, DFU, and Skin Substitute Application
What is an amniotic wound graft? It’s a biologic skin substitute derived from placental tissue — either the amnion alone or combined amnion/chorion layers — applied to chronic or acute wounds to provide growth factors and a scaffolding matrix that stimulates the patient’s own cells to regenerate. It’s not a traditional skin graft; the amniotic tissue typically incorporates or dissolves as healing progresses.
For wound care billing, the key workflow is: document → debride → measure → code.
Your practice needs to establish medical necessity before the first application. That means your chart must show a wound present for at least 4 weeks, a minimum of two conservative treatments attempted (offloading for DFU, compression for VLU, infection management, nutritional support), and serial wound measurements showing failure to progress toward healing (typically defined as less than 50% wound area reduction in 4 weeks).
For more on our complete wound care billing workflow, visit our Wound Care Billing Services page.
Ophthalmic Billing — Dry Eye, Pterygium, and Ocular Surface Procedures
What is the CPT code for pterygium excision with amniotic membrane graft? CPT 65426 (excision of pterygium with graft). If you excise a pterygium and place amniotic membrane as the graft material, bill 65426 — the graft is already described in the “with graft” code. Do not separately bill 65778 or 65779 for the amniotic component when it’s part of a pterygium excision.
How to bill amniotic membrane graft for ophthalmology? For Medicare, bill only the CPT code (65778, 65779, or 65780) — the product cost is wrapped into the procedure reimbursement. Some commercial payers allow separate billing of V2790 for the amniotic device, but verify payer-specific policy before doing so. Submitting V2790 on a Medicare claim for these procedures will result in a denial.
Podiatric and Orthopedic Amniotic Graft Billing
For podiatric foot wounds, use CPT 15277 (first 100 sq cm) and 15278 (each additional 100 sq cm), paired with the appropriate Q-code. Diabetic foot ulcer claims require modifier Q8 in addition to standard laterality modifiers.
For orthopedic tendon wraps, the billing path is murkier. CPT 20926 covers “tissue grafts, other than autograft” and may apply when no other specific code exists. CPT 20999 (unlisted procedure) is a last resort that requires manual review and extensive documentation justifying why no existing code adequately describes the procedure.
⚠️ Warning: Amniotic fluid injections (Q4263) for joints are flagged as “experimental and investigational” by several major payers including Aetna, Cigna, and some UHC plans. If your orthopedic practice is billing injections, expect a meaningful denial rate. Build your appeal packet ahead of time with peer-reviewed studies specific to the product and indication — and see our denial management resources for appeal templates.
How to Bill Amniotic Membrane Graft Procedures — Step by Step
You can have every code memorized and still lose revenue if the workflow breaks down before submission. Here’s the complete process, start to finish.
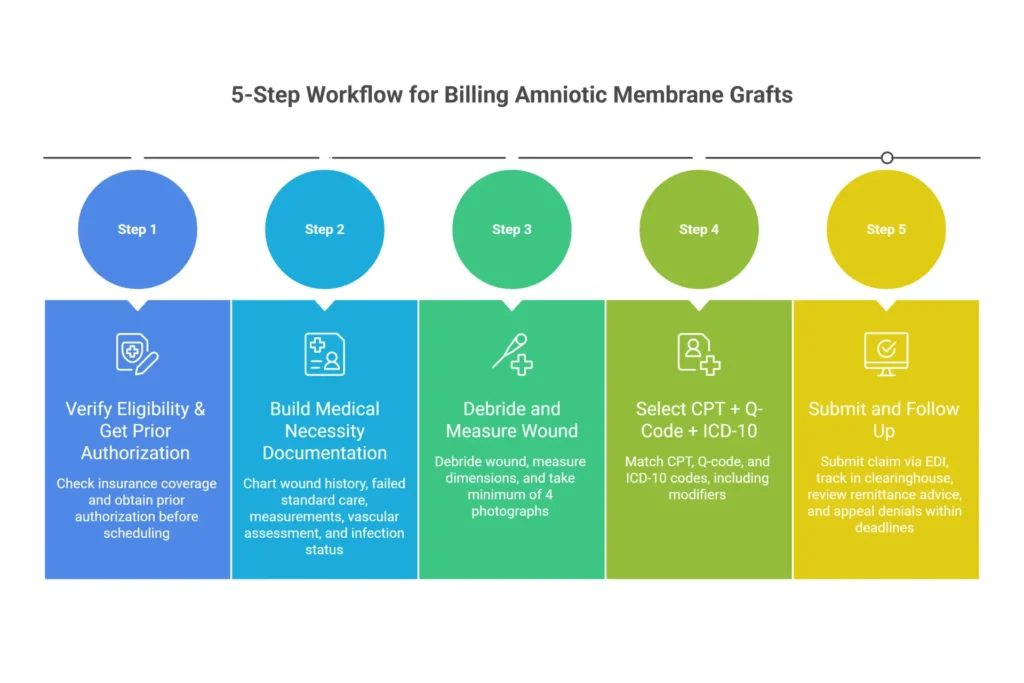
Step 1 — Eligibility & Prior Authorization
Before the patient arrives:
- Verify Medicare Part B active coverage and confirm skin substitute benefit applies
- Check deductible, co-insurance, and out-of-pocket status — especially important if the patient is on a Medicare Advantage plan
- For commercial payers, confirm prior authorization requirements — approximately 80% of plans require it for amniotic grafts
- Submit prior auth with the following bundle: current ICD-10 diagnosis, 4-week wound history with measurements, failed conservative care log (specific treatments, dates, outcomes), product name and Q-code, CPT code for application, and treating provider NPI
Allow 5–14 business days for prior auth processing. Do not schedule the graft application until you have an authorization number in hand.
Step 2 — Documentation Before Service
Build the medical necessity case in the chart before the day of service:
- History of present illness documenting wound duration, etiology, and prior treatment with specific dates
- Serial wound measurements showing inadequate progress (less than 50% area reduction in 4 weeks)
- Vascular assessment for DFU (ankle-brachial index — ABI should be >0.5; if not, document planned vascular referral)
- HbA1c for diabetic patients (many LCDs require documentation; some require HbA1c <12% for coverage)
- Offloading assessment for DFU (type of offloading device prescribed and worn)
- Infection status (no active infection at time of application — document clearly)
- Nutritional status assessment for chronic wound patients
- Informed consent with patient education about amniotic tissue source, FDA HCT/P designation, alternatives, and risks
Step 3 — Measuring Wound Surface Area for Code Selection
How to calculate wound surface area for coding:
- Debride the wound first if indicated — measure after debridement, not before, since debridement often reveals a larger true wound bed
- Place a disposable sterile metric ruler adjacent to the wound (not inside)
- Measure the greatest length in the head-to-toe orientation
- Measure perpendicular width at the widest point (hip-to-hip orientation)
- Multiply: Length (cm) × Width (cm) = Total wound area in cm²
- For irregularly shaped wounds, use the “clock face” or grid method: overlay a sterile transparent grid and count squares, including all squares more than 50% occupied by wound tissue
- Photograph immediately with ruler in frame — timestamp, patient identifier, and date visible
Clinical example: A diabetic patient presents with a plantar foot ulcer measuring 3.8 cm × 2.4 cm. Total wound area = 9.12 cm². You apply a 4×3 cm piece of EpiFix (Q4231), trimming to fit. Bill: CPT 15277 (first 100 sq cm, hands/feet/genitalia) + Q4231 × 9 units (round to nearest whole sq cm, confirm payer rounding policy). Total units billed match graft applied, not graft opened from package.
🩹 Wound Area Calculator & CPT Code Selector
Instantly calculate wound surface area and identify the correct CPT code for amniotic graft billing
⚠️ This tool provides billing references only — not official coding advice. Final code selection must be confirmed by a certified medical coder based on complete procedure documentation, payer policy, and clinical context. Verify all codes against current CMS guidelines.
🔍 Get a Free Wound Care Coding Review from Elite Med Financials →Wound Size Code Selection Table
Table 4: Wound Size and CPT Code Selection Guide — Amniotic Membrane Graft Application
| Total Wound Area | Anatomical Site | CPT Base Code | Add-On Code | Billing Notes |
|---|---|---|---|---|
| ≤25 cm² | Trunk, arms, legs | 15271 | None | First 25 sq cm |
| 26–50 cm² | Trunk, arms, legs | 15271 | 15272 ×1 | One 25 cm² add-on |
| 51–75 cm² | Trunk, arms, legs | 15271 | 15272 ×2 | Two add-ons |
| 76–100 cm² | Trunk, arms, legs | 15271 | 15272 ×3 | Three add-ons |
| >100 cm² | Trunk, arms, legs | 15273 | 15274 per 100 cm² | Switch to 100 cm² base |
| ≤100 cm² | Face, scalp, eyelids, neck | 15275 | None | Includes eyelid wounds |
| 101–200 cm² | Face, scalp, eyelids, neck | 15275 | 15276 ×1 | One add-on |
| ≤100 cm² | Feet, hands, genitalia | 15277 | None | All foot wounds use this |
| 101–200 cm² | Feet, hands, genitalia | 15277 | 15278 ×1 | One 100 cm² add-on |
Step 4 — Selecting CPT + Q-Code + ICD-10 Combination
The three-code combination is your claim’s engine. All three must be consistent with each other and with your procedure note.
For a diabetic foot ulcer:
- CPT: 15277 (feet/hands/genitalia, first 100 cm²)
- Q-code: Q4231 (EpiFix) × 9 units (or whatever your measured area is)
- ICD-10: E11.621 (Type 2 DM with foot ulcer) + L97.419 (non-pressure chronic ulcer of right heel, unspecified severity)
- Modifiers: -RT or -LT, Q8
For a venous leg ulcer:
- CPT: 15271 (trunk/arms/legs, first 25 cm²) or 15273 (larger wounds)
- Q-code: Q4184 (Biovance) × measured units
- ICD-10: I83.011 (varicose veins of right leg with ulcer of thigh)
- Modifiers: -RT or -LT, -59 if bilateral same session
For an ocular surface procedure:
- CPT: 65778 (Prokera, no sutures)
- Q-code: None for Medicare (bundled)
- ICD-10: H16.233 (neurotrophic keratoconjunctivitis, bilateral)
- Modifiers: -50 if bilateral, or separate RT/LT claims
Step 5 — Claim Submission and Follow-Up
- Submit electronically via EDI 837P on CMS-1500 (professional/outpatient)
- Use UB-04 with revenue code 0361 (operating room services), 0624 (medical/surgical supply), or 0636 (drugs) for facility claims
- Track submission confirmation in clearinghouse within 24 hours
- Review the Remittance Advice (RA) within 14–30 days of submission
- Flag any CARC/RARC denial codes immediately for routing to appeals
- For Medicare, Level 1 redetermination deadline is 120 days from denial; build a calendar reminder
For a deeper look at our RCM process including claim scrubbing, submission, and follow-up workflows, visit our Revenue Cycle Management Services page.
Medicare & Insurance Coverage for Amniotic Membrane Grafts
Coverage questions generate more calls to billing departments than almost any other topic. Let’s answer them directly.
Is CPT 65778 Covered by Medicare?
Yes. Medicare covers CPT 65778 for ocular surface disorders under LCD L33941 when all medical necessity criteria are met. Coverage requires a covered diagnosis (neurotrophic keratitis, corneal ulcer, persistent epithelial defect, or severe dry eye), documentation of 4+ weeks of failed conservative therapy, and no contraindications.
Medicare reimbursement for 65778 is approximately $465 (2025 non-facility rate) with the product cost bundled — do not bill V2790 separately for Medicare ophthalmic cases.
Medicare LCD Requirements for Skin Substitutes in Wound Care
Medicare covers wound care skin substitutes (including amniotic membranes) under several MAC-specific LCDs. The most widely applicable:
- L35037 (Novitas Solutions, First Coast Service Options)
- L35086 (Wisconsin Physicians Service / WPS)
- L38640 (Noridian Healthcare Solutions)
- L39217 (CGS Administrators)
Non-negotiable LCD criteria across all MACs:
- Wound present for ≥4 weeks
- Two or more standard conservative treatments failed (offloading + compression or equivalent)
- No active infection, osteomyelitis, or wet gangrene
- Vascular assessment documented (ABI >0.5 for lower extremity wounds)
- Maximum 12 applications per wound site per 12 months
- For applications 5–8: documented continuing progress toward healing
- Applications 9–12: require prior authorization from MAC
Many LCDs also require that wounds be ≤20 cm² for initial approval — larger wounds may require additional documentation or referral.
Payer Coverage Comparison Table
Table 5: Amniotic Membrane Graft Coverage by Payer — 2025–2026
| Payer | Coverage Status | Prior Auth Required | Key Policy Reference | Medical Necessity Standard |
|---|---|---|---|---|
| Medicare FFS | Covered for DFU, VLU, ophthalmic per LCD | Not required (LCD criteria apply) | LCD L35037, L38640, L33941 | 4-week failure, ABI >0.5, ≤12 apps/year |
| Medicare Advantage | Follows original Medicare policy | Often yes (plan-specific) | Plan-specific, same LCD baseline | Same as FFS with plan-specific additions |
| UnitedHealthcare | Covered for DFU, VLU, ophthalmic | Yes (most plans) | MMP105.20 | 4-week failure, ABI >0.5, HbA1c <9% preferred |
| Aetna | Covered where LCD criteria met | Yes | CPB 0244 (ophthalmic), 0293 (wound) | 6-week duration standard (stricter than Medicare) |
| Cigna | Covered with evidence-tier limits | Yes | CP.MP.266 | Tier 1 evidence products only; 12 apps/year cap |
| Anthem/BCBS | State-specific, tiered preferred list | Varies by state plan | State-level policy | Tier 1 products preferred; Tier 2 requires justification |
| Humana | Covered per Medicare LCD | Yes for most | HUM-POLICY-0236 | Mirrors Medicare LCD with commercial add-ons |
| Medicaid FFS | State-by-state; lower reimbursement | State-specific | State PDL/formulary | Usually mirrors Medicare with lower rates |

Commercial Insurance Coverage — What You Need to Know
Most commercial payers follow Medicare’s LCD framework but add layers:
- Tighter evidence requirements: Aetna and Cigna use formal evidence-tier systems. Tier 1 products (strong RCT evidence) like Grafix and EpiFix get through much more reliably than newer or less-studied Q-code products.
- HbA1c restrictions: Some UHC plans deny DFU applications when HbA1c >9%, arguing poor glycemic control undermines healing outcomes. Document glycemic optimization efforts even when A1c is elevated.
- Stricter wound duration requirements: Aetna typically requires 6 weeks of documented failure versus Medicare’s 4 weeks. Check the specific commercial policy before scheduling.
- Preferred product lists: Several commercial payers maintain formularies of covered Q-code products. Using an off-formulary product without prior authorization guarantees a denial.
Is Prokera Covered by Insurance?
Yes, with conditions. Medicare and most commercial payers cover Prokera under CPT 65778 for:
- Neurotrophic keratitis (H16.233)
- Persistent corneal epithelial defects lasting >4 weeks
- Corneal ulcers unresponsive to standard therapy
- Severe dry eye (DEWS Stage 3/4) with documented failed step therapy
- Chemical or thermal burns of the corneal surface
BioTissue, Prokera’s manufacturer, maintains a helpful payer verification program that can accelerate prior authorization processing for new patients. For repeat patients, your billing team should maintain a running prior auth log with expiration dates — most ophthalmic prior auths expire after 90 days.
🔵 Are Amniotic Graft Denials Eating Into Your Revenue?
Most practices never recover the full revenue they lose to preventable claim denials. Our denial management team specializes in amniotic graft appeals, working with Medicare MACs and commercial payers to recover denied claims — and implementing workflow changes to prevent the same denials next time.
Schedule a Free Billing Audit with Elite Med Financials →
Documentation Requirements for Amniotic Graft Billing
In an era of RAC audits and increased OIG scrutiny on skin substitute claims, documentation isn’t a back-office concern — it’s the difference between keeping your revenue and writing a refund check. Here’s exactly what auditors expect to see.
Clinical Documentation Checklist
📋 Amniotic Graft Billing Documentation Checklist
Interactive checklist + full downloadable version — check off items before submission or print for your team
Section A — Before First Application (Medical Necessity)
- ☐ Wound duration established — date of onset or first documented visit clearly noted
- ☐ 4+ weeks of standard wound care documented with specific dates — offloading for DFU, compression for VLU, infection control, debridement schedule
- ☐ Serial wound measurements showing failed progress — less than 50% area reduction over 4 weeks documented
- ☐ Vascular assessment completed (ABI/TBI) — ABI >0.5 for lower extremity wounds; document vascular referral if <0.5
- ☐ HbA1c documented (diabetic patients) — test date and value; most payers require documentation even if A1c is elevated
- ☐ Infection status confirmed — no active infection — explicit statement “no clinical signs of infection” or “infection resolved as of [date]”
- ☐ Nutritional assessment documented — albumin/prealbumin for chronic wound patients; note any interventions
- ☐ Prior authorization obtained (commercial payers) — authorization number, approval date, and expiration date on file
- ☐ Signed informed consent on file — includes amniotic tissue source, FDA HCT/P status, alternatives, and risks
Section B — At Time of Service (Procedure Day Documentation)
- ☐ Wound measured POST-debridement — always after debridement; wound often expands once necrotic tissue is removed
- ☐ Length × Width surface area calculated and documented — exact numeric value in cm² in procedure note; no approximations
- ☐ Wound bed description in procedure note — percentages of granulation, slough, fibrin, and necrosis (should total 100%)
- ☐ Debridement documentation (if performed) — tissue type removed, instrument used, depth reached, and method (sharp/enzymatic/mechanical)
- ☐ Product details recorded at point of care — brand name, Q-code, lot number, expiration date, product package size, and trimmed-to size
- ☐ Units applied match wound measurement — e.g., “9.12 cm² of EpiFix (Q4231) applied” — billed units = applied area, not package size opened
- ☐ Fixation method documented — e.g., “secured with Steri-Strips and non-adherent foam dressing” or “sutured with 4-0 nylon”
- ☐ 4 wound photographs taken with disposable metric ruler visible — pre-debridement, post-debridement, depth/cross-section, post-graft application
- ☐ Photos timestamped and stored in EHR with visit note — patient identifier visible in image or in metadata
Section C — Product Documentation (Q-Code Compliance)
- ☐ Product invoice on file — shows acquisition cost per unit; required for RAC audit defense
- ☐ Lot number and expiration date recorded — photograph the product label and store with the visit documentation
- ☐ FDA 361 HCT/P registration confirmed — manufacturer’s registration certificate on file for each product line used
- ☐ AATB (tissue bank) accreditation confirmed — American Association of Tissue Banks certification for the tissue processing facility
- ☐ Chain of custody documented — receipt date, storage temperature log, and application date all recorded
- ☐ Package insert or HCT/P tracking card retained — 10-year retention required per 21 CFR 1271.270
Section D — For Applications 5 and Beyond
- ☐ Evaluation of wound response to previous applications — serial measurement table showing healing trajectory
- ☐ Photographs showing progressive healing trend — comparison photos at each visit are your strongest appeal evidence
- ☐ Continuing medical necessity attestation — explicit statement justifying continued graft use
- ☐ Modifier -KX appended (applications 5–8) — signals to Medicare that LCD requirements for continued use have been met
- ☐ Prior authorization obtained (applications 9–12) — some MACs require MAC-level prior auth beyond 8 applications
Section E — Claim Submission Quality Check
- ☐ CPT code verified against anatomical site — Foot/hand → 15277 | Trunk/leg → 15271/15273 | Face/scalp → 15275 | Eye → 65778/65779/65780
- ☐ Q-code matches exact product on invoice — cross-reference before submission; payers audit invoices vs. Q-codes
- ☐ ICD-10 codes fully specified (no unspecified codes) — DM first (E11.621) + ulcer second (L97.4xx); never L89.90 “unspecified”
- ☐ Correct modifiers appended — -RT/-LT (laterality), Q8 (DFU), -KX (apps 5–8), -59 (distinct sites), -25 (separate E/M)
- ☐ Claim submitted within timely filing window — Medicare: 12 months | Most commercial: 90 days from date of service
- ☐ Remittance advice reviewed within 30 days — flag any CARC/RARC denial codes immediately for appeal routing
- ☐ All records retained per 21 CFR 1271.270 — HCT/P tracking records: 10-year retention requirement
Wound Measurement & Photo Documentation
The rule every wound care practice should post on the wall: If it’s not photographed and measured, it didn’t happen in an audit.
RAC auditors now specifically look for:
- High-resolution photographs (no blurry phone pics) with a disposable metric ruler visible and adjacent to the wound (not inside it)
- Patient identifier or date of birth visible in the photo, or documented in photo metadata
- Date stamp visible (some EHRs timestamp automatically; confirm this in your system)
- Four standard angles: overall wound context, close-up with ruler (longest dimension), cross-sectional depth if measurable, post-graft application
- Images stored in the EHR with the corresponding visit note
Some practices use standardized wound documentation apps (WoundMatrix, WoundRounds, MolnlyckeCarePath) that timestamp and geotag images automatically — a worthwhile investment if wound care volume is significant.
Product Documentation for Q-Code Billing
For every amniotic graft application, your records must contain:
- Product invoice showing acquisition cost per unit
- Lot number and expiration date (directly from packaging — photograph the label)
- Manufacturer’s FDA 361 HCT/P registration certificate
- AATB (American Association of Tissue Banks) accreditation confirmation
- Chain of custody: receipt date, storage conditions, application date
- Package insert or HCT/P tracking card
Retention requirement: Per 21 CFR 1271.270, all HCT/P tracking records must be maintained for at least 10 years post-administration. This is longer than typical HIPAA retention — build your document retention policy around this requirement.
For additional guidance on wound care documentation requirements, see our detailed Wound Care Billing Guidelines resource.
ICD-10 Codes for Amniotic Membrane Graft Procedures
Diagnosis codes do more than satisfy a form requirement — they establish medical necessity, trigger coverage policies, and determine whether your claim routes to automatic approval or manual review. Specificity is mandatory; unspecified codes are among the top denial triggers.
Primary Diagnosis Codes (Wound Care & Ophthalmic)
Table 6 (partial — see full list below): ICD-10 Reference — Amniotic Membrane Graft Billing
| Condition | ICD-10 Code | Sequence | Critical Notes |
|---|---|---|---|
| Type 2 DM with foot ulcer | E11.621 | Primary | Always sequence DM code first; add ulcer code second |
| Type 1 DM with foot ulcer | E10.621 | Primary | Distinguish Type 1 from Type 2 explicitly in chart |
| Type 2 DM with other skin ulcer | E11.628 | Primary | For non-foot DM ulcers |
| Venous leg ulcer, right calf, no inflammation | I83.011 | Primary | Specify anatomical site and inflammation status |
| Venous leg ulcer, left ankle, with inflammation | I83.023 | Primary | Laterality required |
| Pressure ulcer, sacral region, Stage 3 | L89.039 | Primary | Never use L89.90 (unspecified) — automatic denial trigger |
| Pressure ulcer, right heel, Stage 4 | L89.154 | Primary | Specify side and stage |
| Corneal ulcer, unspecified | H16.009 | Primary | For 65778 medical necessity (specify laterality if known) |
| Neurotrophic keratopathy, bilateral | H16.233 | Primary | Strong medical necessity support for Prokera |
| Dry eye syndrome, bilateral | H04.123 | Primary | DEWS staging must be in documentation |
| Pterygium, right eye | H11.001 | Primary | Use with 65426, not 65778 |
| Chemical burn of cornea, initial encounter | T26.60XA | Primary | Include 7th character A, D, or S |
| Bullous keratopathy | H18.52 | Primary | Supports 65780 reconstruction |
| Stevens-Johnson syndrome | L51.1 | Primary | Supports 65780; document extent of ocular involvement |
ICD-10 Aftercare Codes (Z48.xx) — When and How to Use
What is the ICD-10 code for aftercare skin graft? Z48.817 — Encounter for surgical aftercare following surgery on the skin and subcutaneous tissue.
When to use aftercare codes in ICD-10? Use aftercare codes when:
- The initial wound treatment is complete and the patient returns for routine healing monitoring
- You are performing dressing changes, suture/staple removal, or routine graft site checks
- No active complication or new wound treatment is occurring
Do not use aftercare codes when:
- The wound is still being actively treated with debridement, new graft applications, or antibiotics
- A complication has developed (graft failure, infection, dehiscence) — use the complication code instead
- The patient is presenting for a new problem at the same visit
| Code | Description | Use Case |
|---|---|---|
| Z48.817 | Surgical aftercare, skin and subcutaneous tissue | Standard skin/wound graft follow-up |
| Z48.810 | Surgical aftercare, sense organs | Post-65778/65779 ophthalmic follow-up |
| Z48.01 | Change/removal of surgical dressing | Dressing changes without active wound treatment |
| Z48.02 | Change/removal of wound packing | Deeper wound packing changes |
| Z94.89 | Other transplanted tissue status | Indicates active amniotic graft in situ |
ICD-10-PCS Codes for Amniotic Graft Procedures (Inpatient)
For inpatient hospital encounters (facility billing on UB-04 using ICD-10-PCS):
- 0HRXXXJ — Replacement of skin with synthetic substitute (specify body part, approach, qualifier)
- 0HUXxJZ — Supplement integumentary system, non-autologous tissue substitute
- 08CM0ZZ — Repair right cornea, open approach
- 08CN0ZZ — Repair left cornea, open approach
- 08RM07Z — Replacement of right cornea with autologous tissue substitute (for reconstruction)
These codes apply only to inpatient facility claims. Professional services in the inpatient setting still use CPT codes on the professional claim component.
Common Billing Errors & Denial Management for Amniotic Graft Claims
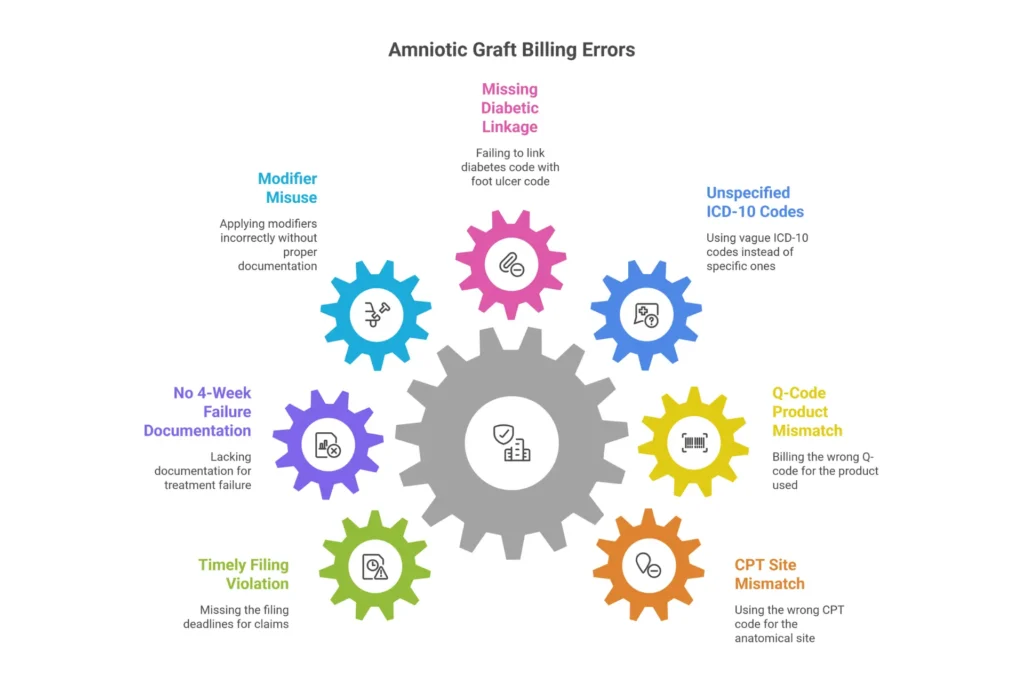
When 25–35% of your amniotic graft claims are getting denied, you need to know exactly where the leaks are. Based on patterns across hundreds of practice audits, here are the errors doing the most damage.
Top 7 Billing Errors That Cause Amniotic Graft Claim Denials
Error #1 — Anatomical site CPT mismatch (accounts for ~35% of code-related denials) Using 65778 (ocular code) for a skin wound, or using 15271 (trunk/extremity code) for a foot wound. The CPT must match the documented anatomical site — and payers validate this against the procedure note. Fix: train your coders to check the CPT-to-site mapping against the procedure note before submission.
Error #2 — Q-code product mismatch (28% of supply-related denials) Billing Q4231 when you used Q4188. Payers routinely cross-reference Q-codes against invoices during audits. The exact Q-code must match the exact product used. Fix: create a product-to-Q-code reference sheet at every clinical workstation and require nursing staff to document the product name and lot number in real time.
Error #3 — Unspecified ICD-10 codes (contributes to ~40% of medical necessity denials) Billing L89.90 (pressure ulcer, unspecified) instead of L89.154 (Stage 4 heel, right). Payers auto-reject unspecified codes for skin substitute claims. Fix: build specificity into your documentation templates so coders never have to guess.
Error #4 — Missing diabetic causal chain Billing L97.419 (foot ulcer) without E11.621 (DM2 with foot ulcer). For diabetic ulcers, diabetes must be sequenced as the primary diagnosis. Fix: require dual-diagnosis coding for all wounds in diabetic patients.
Error #5 — Modifier misuse (-59 without distinct site documentation) Using -59 to “unbundle” debridement from graft application on the same wound site. -59 is appropriate only for distinct anatomical sites. Fix: document separately when procedures involve different wound sites.
Error #6 — No 4-week failure documentation (single largest driver of medical necessity denials — ~45%) The chart shows wound treatment but doesn’t demonstrate that standard care failed. Fix: use a wound progress tracking form that explicitly shows wound size at 4-week intervals with notation: “Wound failed to achieve 50% reduction over 4 weeks of [specific treatments].”
Error #7 — Timely filing violations Commercial payers often require submission within 90 days. A missed deadline is an absolute, non-appealable denial. Fix: build a 30-day submission deadline into your workflow calendar with automated alerts.
How to Appeal a Denied Amniotic Graft Claim
Medicare appeal process:
Level 1 — Redetermination (MAC level, 120-day deadline) Review the RA for denial codes. CARC 96 = non-covered charge, CARC 97 = contractual adjustment (indicates coverage issue), CARC B7 = provider not credentialed. Compile: complete procedure note, serial wound photos with rulers, wound measurement table, failed conservative care log, product lot documentation. Submit via MAC online portal or certified mail. Include a cover letter citing the specific LCD criteria you satisfy.
Level 2 — Reconsideration (QIC level, 180-day deadline) This is your last opportunity to add new evidence. Include peer-reviewed literature supporting the product you used — MiMedx EpiFix clinical trial data (PMID 29262077), Organogenesis studies, or product-specific RCT references. QIC overturn rates are modest (~12%), but the documentation you submit here follows the case through higher levels.
Level 3 — ALJ Hearing (Administrative Law Judge) Well-documented cases have ~80% overturn rates at ALJ, but expect 12–24 month wait times. Focus your ALJ cases on high-dollar denials with clear LCD compliance.
Commercial payer appeals:
- Follow the payer’s specific appeal timeline (usually 60–180 days from denial)
- Attach clinical literature specific to the brand and indication
- Request a Peer-to-Peer review with the payer’s medical director
- Escalate to Independent Review Organization (IRO) if internal appeals fail
- For systematic denials, file formal grievance with the State Insurance Commissioner
Our AR management and denial management team handles appeals for wound care and amniotic graft claims at all payer levels — contact us if your denial backlog needs attention.
Modifier Quick Reference — What Is the Modifier for Skin Graft?
| Modifier | Name | When to Use for Amniotic Grafts |
|---|---|---|
| -59 | Distinct Procedural Service | Two procedures at separate wound sites, same day |
| -51 | Multiple Procedures | Second or subsequent procedures same session |
| -RT / -LT | Right / Left Side | Laterality — required for all foot and eye procedures |
| -50 | Bilateral Procedure | Both eyes same session (ophthalmic claims) |
| -25 | Significant Separate E/M | E/M service on same day as procedure |
| -KX | Requirements Met | Applications 5–8; documents continued healing progress |
| -JZ | Zero Waste (BLA products) | No discarded product — 2026 mandate for BLA biologics |
| Q7 | One Qualifying Chronic Condition | Non-DFU single ulcer |
| Q8 | Two Qualifying Conditions | Required for DFU claims — diabetic + foot ulcer |
| Q9 | Three Qualifying Conditions | Pressure ulcer severity documentation |
Amniotic Membrane Graft Companies, Costs & Reimbursement Rates
Understanding the market doesn’t just satisfy curiosity — it directly affects your practice’s profitability. The 2026 flat-rate transition changes the math considerably.
How Much Does an Amniotic Membrane Graft Cost?
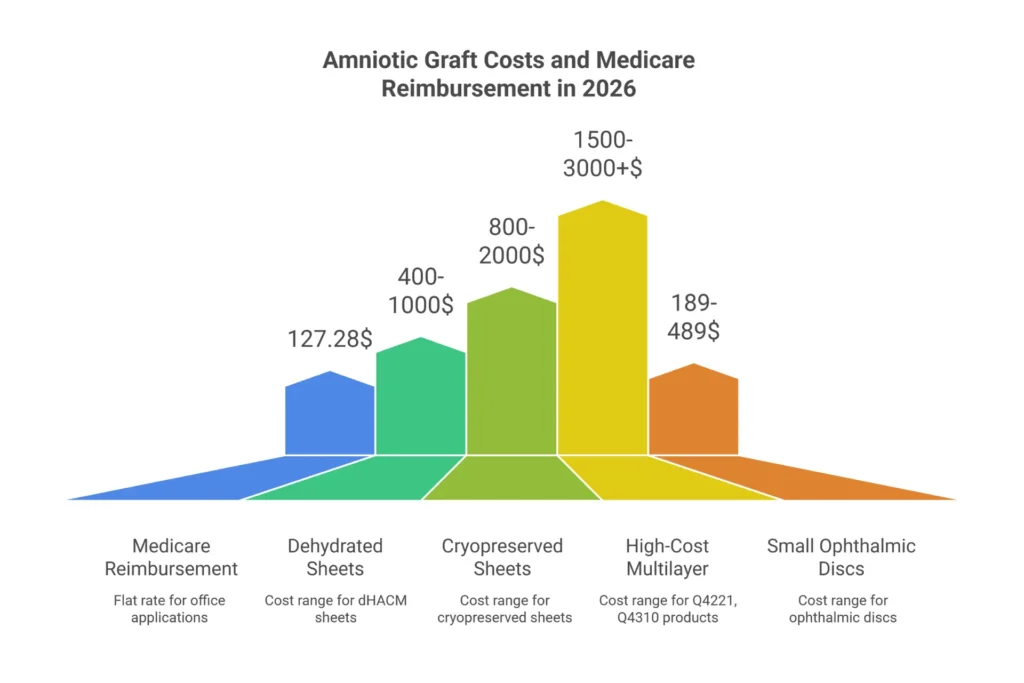
Wholesale acquisition cost (WAC) to providers varies dramatically by product type:
- Small ocular disks (Prokera, similar): $189–$489 per unit
- Dehydrated sheet grafts (dHACM): $400–$1,000 per sq cm
- Cryopreserved sheet grafts: $800–$2,000 per sq cm
- Flowable/injectable amniotic products: $295–$800 per dose
- High-cost multilayer products (Q4221, Q4310): $1,500–$3,000+ per sq cm
Total procedure cost to payers, including product, professional fee, and facility component, typically ranges $1,500–$7,000 per application.
The 2026 margin reality: Medicare’s flat rate of $127.28/cm² under MPFS means profitability at the practice level requires WAC below approximately $95/cm² to maintain a workable margin. Pre-2026, practices routinely saw 30–60% margins. That era is largely over for office-based Medicare cases — making vendor negotiation and site-of-service analysis more important than ever.
Top Amniotic Membrane Graft Companies & Products
Table 7 (partial): Amniotic Membrane Graft Company and Product Reference
| Company | Key Products | Q-Codes | Primary Use Case | Approx. WAC | Est. Reimbursement Range | Notes |
|---|---|---|---|---|---|---|
| MiMedx | EpiFix, AmnioFix | Q4231, Q4238 | DFU, wound, orthopedic | $800–$1,500/cm² | $1,000–$3,400/cm² | 28% market share; strong RCT portfolio |
| Organogenesis | Apligraf, PuraPly AM | Q4173 | Chronic wounds | $650–$1,200/cm² | $650–$1,500/cm² | 18% market share |
| BioTissue | Prokera, AmnioGraft | V2790 (tracking) | Ophthalmology | $400–$800/unit | Bundled in 65778 | Ophthalmic market leader |
| Smith+Nephew | Grafix Core, Grafix PL | Q4133 | DFU, VLU | $1,000–$1,500/cm² | ~$1,200/cm² | Strong Level I evidence |
| Tides Medical | Amnioeffect | Q428x series | Wound care | $600–$1,200/cm² | $800–$1,500/cm² | Emerging market share |
| Sanara MedTech | Amnioforce | Q4238 | Burns, pressure ulcers | $1,200–$2,000/cm² | $1,500–$3,400/cm² | Growing clinical evidence base |
| Katena/Amsler | Generic amnion products | Various Q41xx | Ocular, wound | $400–$600/cm² | $500–$800/cm² | Budget-tier option |
Reimbursement vs. Product Cost — Is It Worth It for Your Practice?
Honestly, the answer in 2026 is: it depends, and you need to model it per site of service.
Office-based (MPFS) math: At $127.28/cm² flat rate, a 20 cm² application generates $2,545.60 in product reimbursement. If your WAC is $80/cm², your product cost is $1,600 — leaving $945.60 plus the separate procedure fee (~$250–$350). That’s a workable case.
If your WAC is $150/cm², you’re paying $3,000 for $2,545.60 in reimbursement on the product alone. You’re losing money before the physician is paid.
Hospital Outpatient (OPPS) math: OPPS APC payments for skin substitutes remain in the $1,850–$3,200 range for many products — potentially offering better margins for complex cases. But HOPD billing has its own compliance requirements, and not every practice has access to that setting.
Strategies for maintaining profitability in 2026:
- Negotiate tiered pricing with multiple vendors — competition drives prices down
- Audit your product formulary: are you using a $2,000/cm² product when a $600/cm² product has equivalent evidence for the indication?
- Model cases by site of service before shifting protocols
- Bill debridement separately when separately performed and separately documented (97597/97598 for selective debridement)
- Track your cost-per-healed-wound metric — total product cost divided by healed outcomes — to evaluate real ROI
Want help modeling amniotic graft profitability for your specific payer mix? Our end-to-end financial management team includes revenue analysis and contract negotiation support.
Frequently Asked Questions — Amniotic Membrane Graft Billing
How do you bill for an amniotic membrane graft?
Bill using CPT 15271–15278 for wound care applications or CPT 65778–65780 for ophthalmic applications. Pair with the product-specific HCPCS Q-code (e.g., Q4231 for EpiFix) billed in units of square centimeters applied. Measure wound area post-debridement (L × W = cm²), document 4+ weeks of failed conservative care, and submit CMS-1500 with lot numbers, photographs, and appropriate ICD-10 diagnosis codes.
Can you bill 15004 and 15275 together?
Generally, no. Surgical preparation (15004) bundles into application codes (15275) per NCCI. You can bill both with modifier -59 only when they’re performed on distinct anatomical sites — document the separate wound sites explicitly.
What is the modifier for skin graft billing?
Primary modifiers: -59 (distinct site), -RT/-LT (laterality), -51 (multiple procedures), -25 (separate E/M), -KX (requirements met for applications 5–8). For DFU claims, add Q8 modifier. For bilateral ophthalmic procedures, use -50 or separate RT/LT claims depending on payer policy.
How to bill CPT 15275?
15275 covers the first 100 sq cm of skin substitute applied to face, scalp, eyelids, or neck. Measure wound post-debridement, bill 15275 for the base, add 15276 for each additional 100 cm², and bill the appropriate Q-code × total cm² applied.
What is the CPT code for amniotic membrane graft on the foot?
CPT 15277 (first 100 sq cm, hands/feet/genitalia). Add 15278 for each additional 100 cm² beyond the first 100. All foot wound applications — dorsal, plantar, heel, toe — use 15277, not the trunk/extremity codes.
What is CPT 65778?
CPT 65778 is placement of amniotic membrane on the ocular surface without sutures — used for self-retaining devices like Prokera for conditions including dry eye, neurotrophic keratitis, and corneal ulcers. Medicare reimburses approximately $465 (2025 NF rate) with a 10-day global period. Product cost is bundled; do not bill Q-codes separately.
What is CPT 65779?
CPT 65779 is placement of amniotic membrane on the ocular surface with sutures. Used for larger or more unstable defects requiring fixation. Approximately $500 Medicare NF reimbursement, 10-day global period.
What is CPT 65780?
CPT 65780 is ocular surface reconstruction using amniotic membrane transplantation — a major procedure for limbal stem cell deficiency, extensive chemical burns, or Stevens-Johnson syndrome. 90-day global period, approximately $1,200 reimbursement.
What is the difference between CPT 65778 and 65779?
Fixation method. 65778 is self-retaining (no sutures, device like Prokera). 65779 requires sutures or glue fixation for larger defects or unstable surfaces. Choose based on the clinical technique used, not the product name.
Does CPT 65778 have a global period?
Yes, 10 days. All post-operative visits within 10 days of the procedure are bundled. Bill separate E/M only for unrelated new problems arising during the global period, with documentation clearly establishing the distinction.
What are Q-codes in billing?
Q-codes are temporary HCPCS Level II codes (Q0000–Q9999) assigned by CMS to identify specific biologic products and supplies — including amniotic membrane grafts — that don’t have permanent CPT or HCPCS codes. For amniotic products, Q-codes identify the specific brand and are billed per square centimeter or milligram applied.
Does Medicare cover Q-codes for amniotic membrane?
Yes. Medicare covers amniotic Q-codes (Q41xx–Q43xx) when billed with appropriate CPT application codes and meeting LCD criteria (L35037, L38640, others). The 2026 flat rate is $127.28/cm² for non-BLA products in office settings.
Is Q4205 a membrane graft or membrane wrap per square cm?
Q4205 (AlloWrap/Stratus) is a membrane wrap per square centimeter, used for circumferential tendon and nerve applications — not flat wound coverage. Bill it differently from sheet grafts like Q4231 (EpiFix).
What Q-code is used for EpiFix?
EpiFix (MiMedx dehydrated human amnion/chorion membrane) bills under Q4231. Bill Q4231 × total square centimeters of graft applied, paired with CPT 15271–15278 based on anatomical site and wound size.
What is the ICD-10 code for aftercare following skin graft?
Z48.817 — Encounter for surgical aftercare following surgery on the skin and subcutaneous tissue. Use for routine post-graft follow-up when no active wound treatment is occurring.
When should you use aftercare codes in ICD-10?
Use aftercare codes (Z48.xx) when the primary surgical treatment is complete and the patient returns for routine monitoring, dressing changes, suture removal, or wound check — without active therapeutic intervention. If the wound still requires active treatment, use the condition code instead.
What is the ICD-10 code for a diabetic foot ulcer?
E11.621 (Type 2 DM with foot ulcer) + L97.4xx (non-pressure chronic ulcer, ankle and midfoot). Sequence E11.621 first — always establish diabetes as the primary code for diabetic wound claims.
Does insurance cover amniotic membrane grafts?
Yes. Medicare covers wound applications per LCD L35037 and ophthalmic use per LCD L33941. Most commercial payers cover amniotic grafts for evidence-supported indications with prior authorization. Approximately 80% of commercial plans require prior auth before the procedure.
Is Prokera covered by Medicare?
Yes, under CPT 65778 for covered indications including neurotrophic keratitis, corneal ulcers, and persistent epithelial defects. The product cost is bundled into the CPT payment — do not submit V2790 separately on Medicare claims.
How much does an amniotic membrane graft cost?
Provider acquisition cost ranges from $189–$489 for small ocular devices to $400–$2,000+ per sq cm for wound care products. Total procedure cost to payers ranges $1,500–$7,000 including product, professional, and facility components. Medicare’s 2026 flat rate is $127.28/cm² for non-BLA products under MPFS.
How long does amniotic membrane graft take to heal?
The graft itself incorporates or dissolves in 7–14 days. The underlying wound typically closes over 4–8 weeks with serial applications. Clinical trials show EpiFix achieving 62% DFU healing by week 12, vs. 8% in standard care controls (PMID 29262077).
How long do you leave an amniotic membrane on the eye?
A self-retaining device like Prokera is typically left in place 3–7 days, dissolving gradually as it releases anti-inflammatory proteins. Any remaining membrane fragments are gently removed at the follow-up visit.
What are the side effects of amniotic membrane for dry eyes?
Mild side effects include foreign body sensation, temporary blurred vision, tearing, and minor discomfort. Rejection is not a concern — amniotic tissue is immunologically privileged. Infection is rare (<1%). Most symptoms resolve within 1–2 weeks post-placement.
What is the most common reason amniotic graft claims are denied?
Missing or inadequate documentation of 4+ weeks of failed conservative care — accounts for approximately 45% of medical necessity denials. The second most common: unspecified ICD-10 codes (~40% of related denials).
How do I appeal a denied amniotic membrane graft claim?
For Medicare: submit Level 1 Redetermination within 120 days with complete procedure note, serial wound photos, wound measurement table, failed care documentation, and product lot records. For commercial payers: follow plan-specific appeal timeline (usually 60–180 days), add peer-reviewed literature for the specific product and indication, and request Peer-to-Peer review if available.
How many amniotic graft applications does Medicare cover per year?
Medicare LCDs typically allow up to 12 applications per wound site per 12 months. Applications 5–8 require documentation of continuing progress toward healing (modifier -KX). Applications 9–12 may require prior authorization from the MAC.
What CPT code do I use for an amniotic membrane graft on the foot?
For a foot wound, use CPT 15277 for the first 100 square centimeters of skin substitute application. Add CPT 15278 for each additional 100 square centimeters beyond the first. Pair with the appropriate Q-code for the specific amniotic product used, billed per square centimeter applied.
How do I know which Q-code to use for amniotic membrane billing?
Match the Q-code to the specific brand you applied. For example, EpiFix uses Q4231, Grafix Core uses Q4133, and Biovance uses Q4184. Each manufacturer’s product has an assigned Q-code — bill the exact code for the exact product, confirmed against the product invoice. Never estimate or substitute a similar Q-code.
What documentation do I need before billing an amniotic graft for Medicare?
You need four weeks of documented standard wound care showing the wound failed to progress, serial wound measurements taken before the graft application, a vascular assessment for lower extremity wounds, infection status documentation confirming no active infection, and the product lot number and expiration date from the graft packaging. Wound photographs with a ruler visible in the image are strongly recommended.
Does Medicare pay for amniotic membrane grafts for wound care?
Yes, Medicare covers amniotic membrane grafts for wound care under Local Coverage Determinations like LCD L35037, when the wound has been present for at least four weeks, standard conservative treatments have failed, there is no active infection, and vascular assessment confirms adequate blood flow. Coverage is limited to a maximum of twelve applications per wound site per year.
What is the difference between CPT 65778 and 65779 for amniotic membrane procedures?
CPT 65778 is used when the amniotic membrane is placed on the eye surface without sutures, using a self-retaining device. CPT 65779 is used when the membrane must be sutured or glued in place because the eye surface requires fixation. The difference is in how the graft is secured, not which product is used.
How do you calculate wound area for amniotic graft billing?
Measure the wound length at its greatest dimension in the head-to-toe direction and the width at the widest point perpendicular to length. Multiply length times width to get the area in square centimeters. Always measure after debridement, not before. Bill Q-code units based on the area of graft applied, not the area of the package opened.
What is the prior authorization process for amniotic membrane grafts with commercial insurance?
Submit a prior authorization request to the payer before the procedure. Include the patient’s diagnosis code, wound duration and measurements, documentation of four or more weeks of failed standard treatment, the specific product name and its Q-code, the CPT application code you plan to use, and the ordering provider’s NPI. Most commercial payers respond within five to fourteen business days.
Is a referral required for amniotic membrane graft procedures?
Referral requirements depend on the patient’s insurance plan type. Traditional fee-for-service Medicare does not require a referral. Medicare Advantage plans, HMO plans, and some PPO plans may require a referral from the primary care provider to a wound care specialist or ophthalmologist before coverage applies. Verify plan requirements at the time of eligibility verification.
What happens if an amniotic graft claim is denied for medical necessity?
You can appeal the denial. For Medicare, file a Level 1 Redetermination with the MAC within 120 days. Include the complete procedure note, wound photographs, the four-week failed conservative care log, wound measurement documentation, and a cover letter citing the specific LCD criteria your patient meets. Commercial payer appeals follow similar processes with payer-specific deadlines.
Can you bill for debridement and amniotic membrane graft on the same day?
Yes, in most cases. Selective debridement codes like 97597 or 97598 can be billed on the same day as graft application codes when debridement is performed as a separate, documented procedure. However, surgical preparation code 15004 is bundled with application codes per NCCI and generally cannot be billed on the same day for the same wound site.
Conclusion: Your Amniotic Graft Billing Strategy for 2026
Amniotic membrane graft billing rewards precision. When you have the right CPT code, the right Q-code, the right diagnosis sequence, and the right documentation in place, these claims move through adjudication cleanly. When any piece is missing or mismatched, you’re looking at a denial — and in many cases, a claim that takes three times as long to work as it would have taken to get right the first time.
Five things to take from this guide:
- CPT code selection is site-specific. The wrong code for the anatomical site is an automatic denial — check the code family against your procedure note before submission on every claim.
- Q-codes identify the exact product. Match the Q-code to the brand invoice, bill per square centimeter applied (not opened), and document the lot number.
- Medical necessity documentation is non-negotiable. Without a documented 4+ week record of failed conservative care, even a perfectly coded claim will be denied. Build that documentation before the first graft goes on.
- The 2026 flat-rate transition changes your profitability math. Model cost-per-application by product and by site of service before committing to a product protocol.
- Denials are recoverable — but only with thorough appeals documentation. Know your MAC, your appeal deadlines, and your supporting clinical literature.
Ready to Stop Losing Revenue to Amniotic Graft Denials?
From CPT code selection to Q-code assignment to Medicare LCD compliance and denial appeals — Elite Med Financials manages your amniotic membrane graft billing end-to-end. Our certified wound care billing specialists handle the complexity so your clinical team can focus on patient outcomes.
We also support your practice with complete medical coding services, credentialing, denial management and AR recovery, and end-to-end revenue cycle management — everything your practice needs to get paid accurately, consistently, and compliantly.
Get a Free Amniotic Graft Billing Audit →
Related Resources from Elite Med Financials
- Complete Wound Care Billing Codes Reference
- Medicare Wound Care Billing Guide
- Wound Care Billing and Coding Services
- Wound Care Billing Guidelines
- Outsource Wound Care Billing Guide
- Outpatient Wound Care Billing
- Wound Care RCM Services
- Wound Care Billing FAQ
- Wound Care Graft Products Program
- Revenue Cycle Management Overview
External References:
- CMS 2026 Medicare Physician Fee Schedule Final Rule — CMS.gov
- AAPC ICD-10/CPT Coding Resources — AAPC.com
This guide was updated March 2026 to reflect CMS 2026 Final Rule changes, including the flat-rate payment transition for non-BLA amniotic products under the Medicare Physician Fee Schedule. Reimbursement rates, LCD criteria, and payer policies change frequently — verify current rates at CMS.gov and confirm coverage policies directly with your MAC and commercial payers before submitting claims.

